抑郁症(专业版)
抑郁症又称抑郁障碍,以显著而持久的心境低落、忧伤和对活动缺乏兴趣等为主要特征。情绪消沉可以从闷闷不乐到悲痛欲绝,或自卑抑郁,甚至悲观厌世、有自杀企图或行为,干扰了患者日常
其他名称:抑郁障碍
英文名称:Depression
抑郁症一般包含以下几种主要类型:
与焦虑、抑郁等神经症相关的一些病因病理研究机制:
有系列的可能增加患抑郁症的风险因素,包括如下:
有关的并发症包括:
综合治疗更有效,选项可包括如下:
调整饮食和生活方式
改变饮食包括如下:
营养与草本综合干预
以下是基于循证医学和循证营养学有关文献综合的结果。
有助于防控抑郁症的营养和草本补充剂,主要包括如下:
1.欧米伽3脂肪酸:
EPA和DHA是神经细胞膜的重要组成部分,影响其流动性和信号传导能力1。它们也在体内被用来产生抗炎代谢物,以及内源性大麻素,一种通过与大麻素受体相互作用来调节神经功能的化合物。这些效果与重度抑郁症的改善有关2,3。临床前研究进一步表明,EPA和DHA可增强神经可塑性4。
适量食用富含脂肪的深海鱼类(EPA、DHA主要膳食来源),可以降低患抑郁症的风险3,5。在患有抑郁症和其他精神疾病的人中,较高水平的ω-3脂肪酸与较轻的症状相关6,7。
大量随机对照试验发现,以2:1或3:1的比例服用EPA和DHA补充剂,每天服用1-2克,对抑郁症有积极影响8,9。欧米伽3脂肪酸也可能对抑郁症和代谢紊乱(如2型糖尿病、肥胖症和心血管疾病)患者有代谢益处10-13。基于大量证据,国际营养精神病学研究学会((ISRPN))建议在治疗和预防严重抑郁症时使用EPA14。还应认识到DHA在支持情绪健康方面的独特作用。DHA是中枢神经系统中最常见的脂肪酸。DHA不仅被整合到神经细胞膜中,这是正常结构和功能所必需的,它也是其他脑细胞的关键组成部分,这些脑细胞利用DHA来支持神经元生长和神经连接的形成。此外,DHA是一种关键的抗炎物质的来源(称为Resolvins和Protectins)3,15。
2. 锌:
锌在体内具有多种功能,包括减少氧化应激、调节神经递质受体功能和调节基因表达16,17。重要的是,锌可以抑制神经元NMDA谷氨酸受体,而NMDA谷氨酸在抑郁症患者中失调,并提高BDNF水平,可促进神经可塑性18,19。对九项研究的荟萃分析发现,低锌摄入与抑郁症风险增加有关20。330此外,据报道,与非抑郁症患者相比,抑郁症患者的锌水平较低,锌缺乏与抑郁症的严重程度和治疗耐药性的增加有关18,21。已经发现,补充锌作为一种独立的治疗方法,与抗抑郁治疗相结合,即使在治疗耐药性抑郁症的情况下,也能改善抑郁症19,22。
一项针对50名超重和肥胖受试者的对照试验发现,抑郁症症状与较低的血液BDNF水平相关,连续12周每天补充30mg锌比安慰剂更有效地提高了BDNF水平并减轻了抑郁症症状23。在30名健康年轻女性中,与单独服用多种维生素相比,在10周内每天补充7毫克锌会降低评估愤怒和敌意的分数24。一项为期12周的试验包括44名患有严重抑郁症的参与者,发现那些除了服用常规的SSRI外,还服用硫酸锌(每天提供25mg元素锌)的人与那些服用SSRI的人相比,抑郁症的改善更大25。在一项对14名重度抑郁症患者进行SSRIs或三环类抗抑郁药治疗的试验中,发现相同剂量的锌在降低抑郁评分方面比安慰剂更有益26。在60例难治性抑郁症患者中,与安慰剂相比,在三环抗抑郁药丙咪嗪(Tofranil)治疗12周后,每天添加25mg锌可获得更快速、更有力的治疗反应27。
3.维生素D:
许多观察性研究注意到低维生素D水平与抑郁症风险之间的联系28-30,一些研究注意到维生素D缺乏与自杀之间的联系31,32。此外,维生素D水平低也可能与抑郁症患者的认知障碍有关33。维生素D与抑郁症之间的联系可能与维生素D的免疫调节抗炎作用、促进神经可塑性和神经发生的能力及其对神经递质水平的影响有关30,34。
一项随机安慰剂对照试验对18,353名50岁及以上的参与者(其中16,657人从未患过抑郁症,1696人患有抑郁症,但至少两年未接受治疗)进行了研究,结果发现在平均5.3年的时间内每天服用2000IU维生素D并没有降低患抑郁症或临床相关抑郁症状的风险35。然而,这项研究的方法和严谨性受到了质疑。305在多项临床研究中,发现维生素D补充剂对抑郁症的有益作用。在临床试验中发现,维生素D可以有效地减轻抑郁症患者的症状,每天服用2800至5600IU,持续1至12个月30。一项对948名重度抑郁症患者进行的四项随机对照试验的荟萃分析发现,补充维生素D对抑郁症症状有实质性的积极影响36。另一项包括9项研究的荟萃分析发现,补充维生素D可以减轻抑郁症症状,改善各种精神病诊断患者的炎症和氧化应激的一些标志物37。
维生素D对健康有更广泛的益处,并可能减轻其他慢性病患者的抑郁情绪。在一项试验中,与12周后接受安慰剂治疗的受试者相比,每天接受4000IU维生素D治疗的2型糖尿病和轻度至中度抑郁症状受试者在情绪和代谢健康指标方面有更大的改善38。一项开放标签试验发现,每天10,000 IU维生素D,持续12个月,可减轻35名多发性硬化症患者的抑郁症状。血液测试显示,研究对象的维生素D水平在研究开始时缺乏或接近缺乏,并在研究过程中变得充足。此外,在整个研究过程中,较高的维生素D水平与较低的抑郁测试分数相关39。
4.益生菌:
肠道微生物组与大脑有着密切的关系,在调节情绪和认知功能方面发挥着重要作用。研究发现,抑郁症患者肠道微生物丰度和多样性降低,这种模式通常与健康状况较差有关40。在一项研究中,食用富含益生菌的发酵食品与患严重抑郁症的风险较低有关41。
多项临床试验、综述和荟萃分析表明,益生菌补充剂可以有效治疗抑郁症42-46。在一项为期8周的试验中,110名患者接受了轻度至中度抑郁症的抗抑郁药物治疗,接受由瑞士乳杆菌R00052和长双歧杆菌R00175组成(Lallemand益生菌专有品牌CEREBIOME®)的100亿CFU的患者在抑郁症测试中的得分比接受安慰剂的患者降低得更多47。这些相同的益生菌菌株以前被证明可以减轻健康志愿者的心理困扰48。一项安慰剂对照试验90名轻度至中度抑郁症受试者发现,在六周内每天服用一种能提供10亿CFU植物乳杆菌补充剂,以及200mg S-腺苷蛋氨酸(SAMe),有效地降低了抑郁症症状评分以及与抑郁症相关的焦虑和认知变化49。在40名重度抑郁症患者中,每天补充20亿CFU的嗜酸乳杆菌、干酪乳杆菌和两歧双歧杆菌、持续8周,可减轻抑郁症状,改善胰岛素抵抗、氧化应激和炎症的一些指标,效果优于安慰剂50。
在一项对40名治疗抵抗性抑郁症患者进行的公开试验中,在标准抗抑郁治疗中每天添加60mg含有丁酸梭菌(Clostridium butyricum)的补充剂,有效率为70%,缓解率为35%51。另一项针对治疗抵抗性抑郁症患者的小型开放试验发现,在抗抑郁治疗中加入一种由嗜酸乳杆菌、两歧双歧杆菌和嗜热链球菌组成的混合益生菌,加上每天1600mg的乳清酸镁,8周后12名参与者中有8名(67%)的抑郁和生活质量评分得到改善52。
益生菌可以减轻其他慢性病患者的抑郁症状。一项包括54名患有2型糖尿病和冠心病的参与者的安慰剂对照试验显示,每天服用嗜酸乳杆菌、罗伊氏乳杆菌、发酵乳杆菌和两歧双歧杆菌各20亿CFU的混合益生菌,以及200 mcg硒,持续12周,可以降低抑郁评分,改善代谢标志物53。同样,一项针对44名心脏病发作后患者的安慰剂对照试验发现,在12周内,每天16亿CFU鼠李糖乳杆菌可以减轻抑郁,减少炎症和氧化应激标志物,提高生活质量54。其他临床试验的结果表明,益生菌可以减轻肠易激综合征患者的抑郁症状55,56。
在一项值得注意的临床试验中,对155名健康人和156名重度抑郁症患者进行了研究,以确定其肠道微生物组和粪便代谢的细菌景观和代谢特征的差异57。试验参与者在三种噬菌体(感染细菌的病毒)、50种粪便代谢物和47种细菌的丰度方面存在显著差异。细菌种类主要分为三个属(分类学类别)之一:拟杆菌属(Bacteroides)、蓝杆菌属(Blautia)和真杆菌属(Eubacterium)。这些属的细菌种类多样,但在体内发挥关键作用,包括调节炎症57,58。重度抑郁症组的拟杆菌丰度显著增加,蓝杆菌属和真杆菌属丰度显著降低。
基于这些发现,作者认为这些属中的某些物种可能是导致重度抑郁症患者常见的炎症失衡的原因。重要的是,这项研究有助于为使用肠道微生物组特征来评估重度抑郁症铺平道路。
5.乙酰L肉碱:
乙酰-L-肉碱是L-肉碱的一种形式,对正常的线粒体能量代谢很重要。乙酰-L-肉碱很容易穿过血脑屏障,增强神经元线粒体代谢59。它被认为具有与抗炎和抗氧化应激机制有关的大脑保护作用,并促进神经可塑性60,61。据报道,抑郁症受试者血液中的乙酰-L-肉碱水平低于非抑郁症受试,并且水平随着抑郁症发病年龄的降低和严重程度的增加而下降;难治性抑郁症和有儿童创伤史的患者的水平最低62。
一项对12项随机对照试验(共791名参与者)进行的荟萃分析发现,乙酰-L-卡尼汀比安慰剂更有效,在减少抑郁症状方面,其疗效与广泛处方的抗抑郁药物相似63。一项对80名患有阈下抑郁症的老年患者进行的为期7周的安慰剂对照试验发现,乙酰-L-肉碱在减轻症状方面与抗抑郁药氟西汀一样有效,但是,乙酰基-L-肉碱组的症状改善比氟西汀更快64。已经发现,每天1500mg的乙酰L肉碱比安慰剂更有效地减轻纤维肌痛综合征患者的抑郁症状和疼痛,并改善生活质量,疗效类似于度洛西汀(Cymbalta)65,66。
6. 维生素B族:
维生素B复合物由九种水溶性B族维生素组成,它们在各种生化反应中起着相关的辅助因子作用,是正常细胞代谢和能量生产所必需的67。正常神经功能需要维生素B1(硫胺素)、维生素B3(烟酸)、维生素B6(吡哆醇)、B9(叶酸)和维生素B12,它们的缺乏与抑郁症有关68。将同型半胱氨酸转化为S-腺苷蛋氨酸(SAMe)需要足够量的三种甲基化酶的B6、B9和B12,SAMe是一种参与合成许多关键生物分子(包括神经递质)的甲硫氨酸。这三种B族维生素中的任何一种缺乏都会提高同型半胱氨酸水平。高同型半胱氨酸水平与抑郁症、心血管、神经系统和其他慢性疾病的风险增加有关69。
临床试验表明,补充B族维生素可以降低健康人的压力水平,改善情绪70。长期补充叶酸和B12可以降低重度抑郁症患者复发的风险,并且可能有助于预防高危人群患上严重抑郁症71。在患有严重抑郁症和导致同型半胱氨酸代谢中断的基因突变的患者中,服用含有代谢形式的B族维生素的多种维生素矿物质补充剂可以降低同型半胱氨酸水平,在减轻抑郁症状方面比安慰剂更有效,8周后缓解率为42%72。在一项为期7天的试验中,发现每天在两餐之间服用200 mg色氨酸、8mg B6和8mg B3的组合比安慰剂更好地减轻症状,该试验包括30名患有严重抑郁症症状但未达到严重抑郁症诊断阈值的年轻人73。
尽管对抑郁症患者的研究结果喜忧参半,但安慰剂对照试验表明,叶酸,特别是甲基叶酸,可能改善抗抑郁治疗的反应74,75。补充维生素B12也被发现可以减轻服用抗抑郁药的患者的症状:在一项包括73名服用抗抑郁药物治疗中重度抑郁症的患者的试验中,在每周肌肉注射1000mcg B12并持续六周的参与者中,100%的人的抑郁评分有20%或更高的改善,而未接受B12注射的参与者中只有69%的人有同样程度的改善76。在一项针对51名严重抑郁症住院患者的安慰剂对照试验中,每天在抗抑郁治疗中添加300mg 维生素B1可以更快地减轻症状77。
7.脱氢表雄酮:
脱氢表雄酮(DHEA)是一种肾上腺激素,可用于产生睾酮和雌二醇。它还具有免疫调节和代谢作用,独立于其作为性激素前体的功能78。DHEA有助于情绪调节,具有神经保护作用,并具有抗应激活性79。抑郁症患者的DHEA-硫酸盐(DHEA-s)水平较低,皮质醇与DHEA-s的比值被认为是衡量抑郁症中应激激素活性的指标80。抑郁症缓解期间皮质醇与DHEA比值的持续紊乱可能是复发风险的指标81。
在一项随机对照试验中,包括17名中年发作的非重度抑郁症参与者,与安慰剂相比,连续三周每天服用90mg DHEA可显著改善抑郁症症状82。同样,一项安慰剂对照试验涉及46名患有严重或非严重中年抑郁症的参与者,该试验也注意到DHEA的抗抑郁作用83。在另一项试验中,22名患有严重抑郁症的参与者(其中一些人正在接受抗抑郁治疗)在六周内每天接受90mg DHEA或安慰剂治疗。与对照组相比,DHEA组中更多的参与者出现了治疗反应,即抑郁症状减少50%84。据报道,DHEA还可以减轻精神分裂症85和HIV/AIDS患者的抑郁症状86。
8. 色氨酸和5-羟色氨酸:
L-色氨酸是肠道和大脑中血清素的氨基酸前体,是肠道和中枢神经系统之间交流的重要介质73,87,88。L-色氨酸也是褪黑素、维生素B3以及其他几种代谢物的前体。色氨酸代谢的改变会增加炎症代谢物的产生,这可能是抑郁症风险的一个因素,并将抑郁症与其他慢性疾病联系起来89-91。一些证据表明,压力会破坏正常的色氨,酸代谢,干扰血清素的产生,导致炎症和抑郁风险增加92,93。L-色氨酸水平已被注意到随着年龄的增长而降低94。
一项对研究的荟萃分析发现,重度抑郁症患者的循环色氨酸水平较低95。色氨酸摄入增加与抑郁症状减轻和社交能力增强有关,随机对照试验表明,每天补充3g L-色氨酸可以改善健康成年人的积极情绪96-99。影响色氨酸代谢的遗传因素似乎在一定程度上决定了个体对L-色氨酸酯补充的反应性100。
5-羟色氨酸(5-HTP)是色氨酸代谢的副产物,是血清素的直接前体。补充5-色氨酸而不是L -色氨酸可以避免其他L-色氨酸代谢物的产生,并且可以绕过色氨酸转化为5-羟色胺的限速步骤。随机对照试验发现,每天服用200-300mg 5-HTP比安慰剂更有效,在治疗重度抑郁症方面可与一线抗抑郁药相当101,102。一些证据表明5-羟色胺还可能增加抗抑郁治疗的反应性。5-羟色胺可引起消化不良,但与血清素综合征或严重的不良反应无关103,104。
9. N-乙酰半胱氨酸:
已知N-乙酰半胱氨酸(NAC)是半胱氨酸的衍生物。作为半胱氨酸的来源,它有助于提升谷胱甘肽水平,谷胱甘肽是代谢毒素和减少全身氧化应激所必需的。NAC在大脑中具有重要功能,可减少氧化应激和神经炎症,增强线粒体活性,调节多巴胺和谷氨酸信号传导,促进神经可塑性等重要功能105。
一项针对252名重度抑郁症参与者的对照试验将标准护理加每日2000mg NAC与标准护理加安慰剂进行了比较。在抑郁症最严重的患者中,从第六周开始,NAC的使用使症状得到了统计学上的显著改善,并一直持续到研究结束。此外,在试验结束后四周的随访中,NAC使用者的功能损伤得到了更大的改善106。另一项试验发现,每天1800mg NAC可减轻双相或重度抑郁症患者的抑郁和焦虑症状,C反应蛋白(系统炎症的标志物)基线水平升高107。在一项对35名有非自杀性自伤史的女性青年的试验中,NAC的剂量逐渐增加到每天3600mg,可以减轻抑郁症状和自残行为108。多项临床试验发现,NAC可以减轻抑郁症和其他精神和神经疾病患者的抑郁症状109。
10. S-腺苷蛋氨酸:
S-腺苷甲硫氨酸(SAMe)是合成许多关键生物分子(包括神经递质和激素)所需的单碳甲基的重要来源。从甲硫氨酸(或称蛋氨酸)生产SAMe需要足够量的叶酸和维生素B12,它们在化学反应中起辅助因子的作用110。研究发现,抑郁症患者的SAMe水平降低111。对2005年的研究进行的系统综述以及2003年和1994年发表的两项荟萃分析都发现了支持SAMe抗抑郁作用的证据112-114;然而,最近的试验无法明确证实这些发现111。
在一项包括49名轻度至中度抑郁症患者的安慰剂对照试验中,每天800mg SAMe持续8周,可显著减轻抑郁症症状,尤其是轻度抑郁症患者;然而,由于研究参与者人数较少,安慰剂反应非常强烈,SAMe的改善在统计学上没有显著意义115。同样,在对单独药物反应不足的患者中,将SAMe添加到抗抑郁治疗中,可导致具有临床意义但无统计学意义的改善,因为安慰剂组的反应率为50%116。
2016年,一项对8项试验的系统综述共包括934名受试者,发现SAMe与某些抗抑郁疗法具有相似的疗效,但总体证据质量较差117。一项为期12周的随机对照试验,189名重度抑郁症患者每天服用1600–3200mg SAMe,或安慰剂组发现三者效果相同118;然而,对这些发现的第二次分析显示,SAMe在男性中优于安慰剂,但在女性中则不然119。性别的潜在影响可以部分解释多年来临床试验结果的不一致性。
总体而言,现有证据不足以得出SAMe治疗抑郁症的疗效结论。对服用抗抑郁药的人进行SAMe辅助治疗可能是有效的,但需要更高质量的研究来阐明益处的程度(如果有的话)111。SAMe是双相情感障碍患者的禁忌症,因为据报道它在某些情况下会诱发躁狂120。
11.肌酸:
肌酸存在于肉类和鱼类中。它在调节高活性细胞(如肌肉和脑细胞)的能量产生方面发挥着关键作用。饮食中肌酸摄入量低与抑郁症风险增加有关,大脑肌酸代谢的改变已被确定为抑郁症的潜在因素121,122。
一项针对52名重度抑郁症女性的随机对照试验发现,艾司西酞普兰加肌酸一水合物联合治疗,每天3g,持续一周,然后每天5g,持续七周,与艾司西酞普兰加安慰剂相比,治疗反应性更好123。一项后续研究发现,补充肌酸也会增加大脑中神经连接的形成124。在一项针对15名耐治疗抑郁症女性的公开试验中,在8周内每天添加5g肌酸一水合物加100mg 5-HTP可使抑郁症状评分降低60%125。一项针对5名对氟西汀无反应的重度抑郁症少女的公开试验发现,在8周内向每天添加4g肌酸可使症状评分降低56%126。
然而,一项针对18名三周后对抗抑郁治疗无反应的抑郁症女性的对照试验发现,每天添加5或10g肌酸在提高反应率方面并不比安慰剂更有效127。有必要对肌酸进行进一步的研究,以阐明肌酸是否比安慰剂对重度抑郁症患者有益。
12.肌醇:
肌醇存在于许多动植物食品中。肌醇以磷脂酰肌醇及其衍生物的形式储存在细胞膜中。在中枢神经系统中,肌醇有助于调节神经元信号:神经细胞上神经递质受体的激活刺激肌醇从细胞膜输送到细胞中,在细胞中通过神经递质释放调节信号的持续存在128。据报道,抑郁症患者的脑内肌醇水平较低,并且可能由于成功的治疗而正常化128,129。口服肌醇补充剂可能有助于使大脑肌醇水平正常化128,130。
一项针对28名抑郁症参与者的随机对照试验发现,在为期四周的时间里,每天12g肌醇比安慰剂更有效地减轻症状131。在几个对照试验中,肌醇也被发现比安慰剂略好地减轻抑郁症症状132。然而,研究人员未能发现,与安慰剂相比,在接受SSRI治疗失败的抑郁症患者中,肌醇的益处133,134。
13.镁:
镁在大脑中具有许多重要功能,包括参与神经信号传递的调节135。一些研究报告称,较高的镁摄入量和血清水平与较低的抑郁可能性和抑郁症状的减少有关136,137。一些非对照试验发现,补充镁对减少抑郁症状有效138。在一项包括126名患者的交叉试验中对于轻度至中度抑郁症,在六周内每天补充248mg元素镁(氯化镁)与不治疗相比,症状显著减轻139。尽管大多数安慰剂对照试验发现镁的治疗效果与安慰剂没有什么不同138,一项试验发现,在60名镁缺乏的抑郁症患者中,每天500mg氧化镁持续8周,比安慰剂显著减轻症状140。
人们认为,镁穿过血脑屏障的能力差可能是其在临床试验中表现不佳的原因141。在细胞和动物研究中,苏糖酸镁(一种更容易渗透到中枢神经系统的镁盐)被发现可以提高大脑镁水平,改善大脑结构和功能142-145,在一项针对大鼠的研究中,苏糖酸镁比其他形式的镁更能减轻抑郁样症状,改善记忆力146。
14.牛磺酸:
中枢神经系统是牛磺酸的高聚集区,牛磺酸可以减少大脑氧化应激,刺激神经生长和发育,并参与神经信号传递的调节147。在实验诱发的抑郁症动物的大脑中,牛磺酸、天冬氨酸、甘氨酸和γ氨基丁酸(GABA)的水平都有所下降148。一个研究小组在糖尿病实验动物中发现了牛磺酸的抗抑郁作用,并指出补充牛磺酸可以逆转糖尿病相关的GABA信号传导和BDNF产生的减少,防止脑萎缩并改善记忆149,150。据报道,牛磺酸还能通过调节应激激素水平和促进神经生长、神经存活和新神经的形成,减少暴露于慢性应激的动物的抑郁样症状151。
在一项随机对照试验中,86名因首次精神病发作而接受治疗的患者每天服用4g牛磺酸或安慰剂,同时接受标准药物治疗12周;接受牛磺酸治疗的患者在精神症状(包括抑郁症状)和整体功能方面有更大的改善152。
15.圣约翰草:
多项临床试验表明,圣约翰草(贯叶连翘)可以减轻重度抑郁症的症状,其疗效与一线抗抑郁药物相似,但副作用更少153-155。一项荟萃分析包括35项随机对照试验,共有6,993名参与者,证实了圣约翰草对轻度至中度抑郁症的益处。然而,人们对它在严重的抑郁症中的作用知之甚少156。
圣约翰草中的一些活性化合物对单胺氧化酶有微弱的抑制作用,单胺氧化酶是一种分解神经递质的酶,而其他成分具有抗炎和清除自由基的作用157,158。研究认为其抗抑郁特性与多种活性化合物的协同作用有关159。标准化提取物通常含有0.1-0.3%的金丝桃素(Hypericin)和高达6%的贯叶金丝桃素(Hyperforin)160。
圣约翰草的提取物,尤其是那些富含贯叶金丝桃素的提取物,可以促进肠壁和肝脏的解毒活动,这可能影响许多药物的代谢。因此,服用药物并想要添加圣约翰草的人使用低贯叶金丝桃素制剂是最安全的,并应仔细监测患者服用药物的条件160。
在欧洲,圣约翰草是最常用的抗抑郁药之一,在德国的处方量几乎是所有其他处方抗抑郁药的二倍161。
16.藏红花:
藏红花及其活性成分因其对抑郁症的影响而被广泛研究。其抗抑郁作用可能是由于其抗炎作用、提高多巴胺水平的能力以及与内源性阿片受体的相互作用162。多项对照临床试验和荟萃分析表明,藏红花提取物(通常每天30mg)比安慰剂更有效,在改善轻度至中度抑郁方面与抗抑郁药物相当163-166。一项荟萃分析包括21项对照试验的结果,发现藏红花提取物可以降低自我报告的抑郁和焦虑评分,改善睡眠质量167。
在一项针对40名重性抑郁症患者的安慰剂对照试验中,与单独的抗抑郁治疗相比,抗抑郁治疗加上每天30mg番红花苷(藏红花中的一种活性化合物)持续4周的联合治疗,在减轻症状方面更有效168。在54名2型糖尿病和轻度至中度合并抑郁和焦虑的患者中,与安慰剂相比,每天服用30mg藏红花提取物8周后,情绪症状和睡眠改善更多169。在139名尽管接受了抗抑郁药物治疗但仍患有持续性抑郁症的患者中,添加标准化藏红花提取物,每天28mg,8周后降低了医生评估的抑郁症评分,但没有自我评估评分,优于安慰剂170。
藏红花提取物已被证明可以降低超重女性171、有更年期症状的健康女性172、正在药物滥用康复中的HIV/AIDS患者173、冠状动脉疾病患者174、纤维肌痛综合征患者175的抑郁症状。安慰剂对照临床试验进一步表明,藏红花与抗抑郁药联合使用,可以减轻男性和女性与性功能障碍相关的抗抑郁药副作用176,177。
17.薰衣草:
初步研究表明,薰衣草芳香疗法可以缓解抑郁症状。在一些小型研究中,口服薰衣草制剂如提取物、茶或酊剂,已被证明可以改善抑郁症状178。在一项单盲临床试验中,60名老年受试者被分配每天喝两次薰衣草茶或两周内不接受治疗。情绪评分采用标准化问卷进行评估。与不喝任何薰衣草茶的人相比,薰衣草茶组的抑郁症得分显著下降179。
在一项招募318名患有混合焦虑和抑郁的成年人的试验中,薰衣草提取物(Silexan,薰衣草口服品牌Silexan™)每天80mg、持续治疗70天后,在标准化抑郁评分表上的得分显著高于安慰剂180。在一项单独的开放标签试验中,对报告焦虑和不安的人进行了相同提取物的研究,治疗6周后,抑郁得分比基线显著下降181。
在另一项试验中,45名患有严重抑郁症的成年人被随机分配接受薰衣草酊(50%酒精中的1:5提取物)加安慰剂、薰衣草酊加抗抑郁药丙咪嗪或丙咪嗪加安慰剂治疗四周。研究发现,薰衣草和丙咪嗪的组合在减轻抑郁方面比单独使用丙咪嗪更有效,但单独使用薰衣草的效果不如单独使用丙咪嗪。研究人员得出结论:“这项研究的主要总体发现是,作为辅助治疗,[薰衣草]酊剂可能对轻度至中度抑郁症有治疗益处。”182
18.红景天:
作为适应原,红景天是一种提高应激弹性的疗法,历史上一直被用于治疗疲劳、增强耐力和促进长寿183。临床前证据表明,红景天调节神经递质信号传导并使应激反应正常化。它还具有抗炎和氧化应激减轻作用,这可能有助于其抗抑郁活性,并已被证明在健康志愿者的大脑中抑制抑郁样信号传导184-186。
一项针对89名轻度至中度抑郁症患者的对照试验发现,与安慰剂相比,每天340mg和680mg的红景天提取物在42天后减轻了抑郁症症状。尽管两种剂量的总体效果相似,但注意到接受较高剂量的患者自尊显著改善,而接受较低剂量的患者则没有观察到这一点187。在一项随机对照试验中,100名轻度至中度抑郁症患者接受了SSRI舍曲林加上每天300mg的红景天的治疗,每天600mg的红景天或安慰剂。12周后,接受高剂量红景天治疗的患者症状改善最大188。此外,多项公开试验的结果进一步证明红景天具有有意义的抗抑郁活性189。在一项针对57名轻度至中度抑郁症患者的12周比较试验中,与安慰剂相比,红景天提取物的剂量增加(每天340–1360mg)导致改善的可能性略低于舍曲林,但不良副作用较少186。
19.香蜂草:
香蜂草(Melissa officinalis)在欧洲历来被用于治疗情绪障碍,包括抑郁症和焦虑症,以及管理压力和促进睡眠190。当代临床前研究表明,香蜂草具有一些药理学特性,这可能部分解释了它影响情绪的能力。香蜂草提取物已在体外显示出抑制单胺氧化酶A(MAO-A)的作用,这种作用可以调节参与情绪调节的单胺神经递质的代谢191。
在一项小型试点研究中,45名患有严重抑郁症的成年人被随机分配服用氟西汀、香蜂草或另一种草药英国薰衣草(Lavandula angustifolia)190。名受试者在基线、两周、四周和八周时使用HAM-D评估情绪。在研究过程中,香蜂草和薰衣草在降低受试者HAM-D评分方面与氟西汀一样有效。这项研究的作者承认了这项工作的初步性质:“尽管在我们的研究中,M.officinalis和L.angustifolia的效果与氟西汀相当,但一项荟萃分析显示,这些效果[与安慰剂效应192。关于这一争议,需要更大规模的随访时间更长的试验,包括安慰剂组,以评估这些草药的长期安全性和有效性。”
早期的一项随机安慰剂对照试验评估了香蜂草对80名慢性稳定型心绞痛患者情绪和睡眠障碍的影响。参与者被随机分配接受每天3g香蜂草或安慰剂治疗,为期8周。使用标准化问卷对情绪进行评估。在研究结束时,服用香蜂草的受试者在抑郁、焦虑、压力和睡眠障碍方面的得分与服用安慰剂的受试对象相比显著降低193。另一项小型试验发现,香蜂草有助于缓解失眠者的抑郁情绪。在这项研究中,香蜂草与另一种草药薄荷联合使用,经过四周的治疗,这种组合比安慰剂更能改善情绪相关评分194。
更多内容可点击其个性化综合干预方案如下:
其他抑郁症管理:
以及参与本网如下专文的相关内容:
医疗干预
一般治疗措施包括如下:
心理疗法
对抑郁症的心理治疗由各种类型的咨询组成,如:
参考文献:
1. Zárate R et al. Significance of long chain polyunsaturated fatty acids in human health. Clin Transl Med. 2017;6(1):25.
2. Yang B et al. Clinical Efficacy and Biological Regulations of ω-3 PUFA-Derived Endocannabinoids in Major Depressive Disorder. Psychotherapy and psychosomatics. 2019;88(4):215-224.
3. Messamore E et al. Polyunsaturated fatty acids and recurrent mood disorders: Phenomenology, mechanisms, and clinical application. Prog Lipid Res. 2017;66:1-13.
4. Yu JZ et al. N-3 polyunsaturated fatty acids promote astrocyte differentiation and neurotrophin production independent of cAMP in patient-derived neural stem cells. Molecular psychiatry. 2021 Sep;26(9):4605-4615.
5. Grosso G et al. Dietary n-3 PUFA, fish consumption and depression: A systematic review and meta-analysis of observational studies. J Affect Disord. 2016;205:269-281.
6. Berger M et al. Relationship Between Polyunsaturated Fatty Acids and Psychopathology in the NEURAPRO Clinical Trial. Frontiers in psychiatry. 2019;10:393.
7. Scola G et al. Alterations in peripheral fatty acid composition in bipolar and unipolar depression. J Affect Disord. 2018;233:86-91.
8. Liao Y et al. Efficacy of omega-3 PUFAs in depression: A meta-analysis. Translational psychiatry. 2019;9(1):190.
9. Bozzatello P et al. Supplementation with Omega-3 Fatty Acids in Psychiatric Disorders: A Review of Literature Data. J Clin Med. 2016;5(8).
10. Keshavarz SA et al. Omega-3 supplementation effects on body weight and depression among dieter women with co-morbidity of depression and obesity compared with the placebo: A randomized clinical trial. Clin Nutr ESPEN. 2018;25:37-43.
11. Mazaherioun M et al. Long Chain n-3 Fatty Acids Improve Depression Syndrome in Type 2 Diabetes Mellitus. Iranian journal of public health. 2018;47(4):575-583.
12. Chang JP et al. Omega-3 polyunsaturated fatty acids in cardiovascular diseases comorbid major depressive disorder - Results from a randomized controlled trial. Brain Behav Immun. 2020;85:14-20.
13. Jiang W et al. Long-Chain Omega-3 Fatty Acid Supplements in Depressed Heart Failure Patients: Results of the OCEAN Trial. JACC Heart Fail. 2018;6(10):833-843.
14. Guu TW et al. International Society for Nutritional Psychiatry Research Practice Guidelines for Omega-3 Fatty Acids in the Treatment of Major Depressive Disorder. Psychotherapy and psychosomatics. 2019;88(5):263-273.
15. DiNicolantonio JJ et al. The Importance of Marine Omega-3s for Brain Development and the Prevention and Treatment of Behavior, Mood, and Other Brain Disorders. Nutrients. 2020;12(8).
16. Prasad AS et al. Molecular Mechanisms of Zinc as a Pro-Antioxidant Mediator: Clinical Therapeutic Implications. Antioxidants (Basel, Switzerland). 2019;8(6).
17. Doboszewska U et al. Zinc in the Monoaminergic Theory of Depression: Its Relationship to Neural Plasticity. Neural plasticity. 2017;2017:3682752.
18. Wang J et al. Magnesium, Selenium and Depression: A Review of the Evidence, Potential Mechanisms and Implications. Nutrients. 2018;10(5).
19. Petrilli MA et al. The Emerging Role for Zinc in Depression and Psychosis. Frontiers in pharmacology. 2017;8:414.
20. Li Z et al. Dietary zinc and iron intake and risk of depression: A meta-analysis. Psychiatry research. 2017;251:41-47.
21. Siwek M et al. Serum zinc level in depressed patients during zinc supplementation of imipramine treatment. J Affect Disord. 2010;126(3):447-452.
22. Szewczyk B et al. The role of magnesium and zinc in depression: similarities and differences. Magnesium research. 2018;31(3):78-89.
23. Solati Z et al. Zinc monotherapy increases serum brain-derived neurotrophic factor (BDNF) levels and decreases depressive symptoms in overweight or obese subjects: a double-blind, randomized, placebo-controlled trial. Nutritional neuroscience. 2015;18(4):162-168.
24. Sawada T et al. Effect of zinc supplementation on mood states in young women: a pilot study. European journal of clinical nutrition. 2010;64(3):331-333.
25. Ranjbar E et al. Effects of zinc supplementation on efficacy of antidepressant therapy, inflammatory cytokines, and brain-derived neurotrophic factor in patients with major depression. Nutritional neuroscience. 2014;17(2):65-71.
26. Nowak G et al. Effect of zinc supplementation on antidepressant therapy in unipolar depression: a preliminary placebo-controlled study. Pol J Pharmacol. 2003;55(6):1143-1147.
27. Siwek M, Dudek D, Paul IA, et al. Zinc supplementation augments efficacy of imipramine in treatment resistant patients: a double blind, placebo-controlled study. J Affect Disord. 2009;118(1-3):187-195.
28. Woodward G et al. Serum Vitamin D and Magnesium levels in a psychiatric cohort. Psychiatr Danub. 2019;31(Suppl 3):221-226.
29. Schaad KA et al. The relationship between vitamin D status and depression in a tactical athlete population. Journal of the International Society of Sports Nutrition. 2019;16(1):40.
30. Geng C et al. Vitamin D and depression: mechanisms, determination and application. Asia Pac J Clin Nutr. 2019;28(4):689-694.
31. Kim SY et al. Vitamin D deficiency and suicidal ideation: A cross-sectional study of 157,211 healthy adults. Journal of psychosomatic research. 2020;134:110125.
32. Gokalp G. The association between low vitamin D levels and suicide attempts in adolescents. Ann Clin Psychiatry. 2020;32(2):106-113.
33. Belzeaux R et al. Association between hypovitaminosis D and cognitive inhibition impairment during major depression episode. J Affect Disord. 2018;225:302-305.
34. Grudet C et al. Vitamin D and inflammation in major depressive disorder. J Affect Disord. 2020;267:33-41.
35. Okereke OI et al. Effect of Long-term Vitamin D3 Supplementation vs Placebo on Risk of Depression or Clinically Relevant Depressive Symptoms and on Change in Mood Scores: A Randomized Clinical Trial. JAMA. 2020;324(5):471-480.
36. Vellekkatt F et al. Efficacy of vitamin D supplementation in major depression: A meta-analysis of randomized controlled trials. J Postgrad Med. 2019;65(2):74-80.
37. Jamilian H et al. The effects of vitamin D supplementation on mental health, and biomarkers of inflammation and oxidative stress in patients with psychiatric disorders: A systematic review and meta-analysis of randomized controlled trials. Progress in neuro-psychopharmacology & biological psychiatry. 2019;94:109651.
38. Omidian M et al. Effects of vitamin D supplementation on depressive symptoms in type 2 diabetes mellitus patients: Randomized placebo-controlled double-blind clinical trial. Diabetes Metab Syndr. 2019;13(4):2375-2380.
39. Kotb MA et al. Effect of vitamin D replacement on depression in multiple sclerosis patients. Mult Scler Relat Disord. 2019;29:111-117.
40. Du Y et al. Crosstalk between the microbiota-gut-brain axis and depression. Heliyon. 2020;6(6):e04097.
41. Kim CS et al. Probiotic food consumption is associated with lower severity and prevalence of depression: A nationwide cross-sectional study. Nutrition (Burbank, Los Angeles County, Calif). 2019;63-64:169-174.
42. Sanada K et al. Gut microbiota and major depressive disorder: A systematic review and meta-analysis. J Affect Disord. 2020;266:1-13.
43. Chao L et al. Effects of Probiotics on Depressive or Anxiety Variables in Healthy Participants Under Stress Conditions or With a Depressive or Anxiety Diagnosis: A Meta-Analysis of Randomized Controlled Trials. Front Neurol. 2020;11:421.
44. Amirani E et al. The effects of probiotic supplementation on mental health, biomarkers of inflammation and oxidative stress in patients with psychiatric disorders: A systematic review and meta-analysis of randomized controlled trials. Complementary therapies in medicine. 2020;49:102361.
45. Yong SJ et al. Antidepressive Mechanisms of Probiotics and Their Therapeutic Potential. Front Neurosci. 2019;13:1361.
46. Goh KK et al. Effect of probiotics on depressive symptoms: A meta-analysis of human studies. Psychiatry research. 2019;282:112568.
47. Kazemi A et al. Effect of probiotic and prebiotic vs placebo on psychological outcomes in patients with major depressive disorder: A randomized clinical trial. Clin Nutr. 2019;38(2):522-528.
48. Messaoudi M et al. Assessment of psychotropic-like properties of a probiotic formulation (Lactobacillus helveticus R0052 and Bifidobacterium longum R0175) in rats and human subjects. The British journal of nutrition. 2011;105(5):755-764.
49. Saccarello A et al. Oral Administration of S-Adenosylmethionine (SAMe) and Lactobacillus Plantarum HEAL9 Improves the Mild-To-Moderate Symptoms of Depression: A Randomized, Double-Blind, Placebo-Controlled Study. Prim Care Companion CNS Disord. 2020;22(4).
50. Akkasheh G et al. Clinical and metabolic response to probiotic administration in patients with major depressive disorder: A randomized, double-blind, placebo-controlled trial. Nutrition. 2016;32(3):315-320.
51. Miyaoka T et al. Clostridium butyricum MIYAIRI 588 as Adjunctive Therapy for Treatment-Resistant Major Depressive Disorder: A Prospective Open-Label Trial. Clin Neuropharmacol. 2018;41(5):151-155.
52. Bambling M et al. A combination of probiotics and magnesium orotate attenuate depression in a small SSRI resistant cohort: an intestinal anti-inflammatory response is suggested. Inflammopharmacol. 2017;25(2):271-274.
53. Raygan F et al. The effects of probiotic and selenium co-supplementation on mental health parameters and metabolic profiles in type 2 diabetic patients with coronary heart disease: A randomized, double-blind, placebo-controlled trial. Clin Nutr. 2019;38(4):1594-1598.
54. Moludi J et al. The Effect of Probiotic Supplementation on Depressive Symptoms and Quality of Life in Patients After Myocardial Infarction: Results of a Preliminary Double-Blind Clinical Trial. Psychosomatic medicine. 2019;81(9):770-777.
55. Zhang L et al. Clinical characteristic and fecal microbiota responses to probiotic or antidepressant in patients with diarrhea-predominant irritable bowel syndrome with depression comorbidity: a pilot study. Chin Med J (Engl). 2019;132(3):346-351.
56. Pinto-Sanchez MI et al. Probiotic Bifidobacterium longum NCC3001 Reduces Depression Scores and Alters Brain Activity: A Pilot Study in Patients With Irritable Bowel Syndrome. Gastroenterology. 2017;153(2):448-459.e448.
57. Yang J et al. Landscapes of bacterial and metabolic signatures and their interaction in major depressive disorders. Science advances. 2020;6(49).
58. Mukherjee A et al. Gut microbes from the phylogenetically diverse genus Eubacterium and their various contributions to gut health. Gut Microbes. 2020;12(1):1802866.
59. Pennisi M et al. Acetyl-L-Carnitine in Dementia and Other Cognitive Disorders: A Critical Update. Nutrients. 2020;12(5).
60. Maldonado C et al. Potential Therapeutic Role of Carnitine and Acetylcarnitine in Neurological Disorders. Curr Pharm Des. 2020;26(12):1277-1285.
61. Wang SM et al. A review of current evidence for acetyl-l-carnitine in the treatment of depression. Journal of psychiatric research. 2014;53:30-37.
62. Nasca C et al. Acetyl-l-carnitine deficiency in patients with major depressive disorder. Proc Natl Acad Sci U S A. 2018;115(34):8627-8632.
63. Veronese N et al. Acetyl-L-Carnitine Supplementation and the Treatment of Depressive Symptoms: A Systematic Review and Meta-Analysis. Psychosomatic medicine. 2018;80(2):154-159.
64. Bersani G et al. L-Acetylcarnitine in dysthymic disorder in elderly patients: a double-blind, multicenter, controlled randomized study vs. fluoxetine. Eur Neuropsychopharmacol. 2013;23(10):1219-1225.
65. Leombruni P et al. A randomised controlled trial comparing duloxetine and acetyl L-carnitine in fibromyalgic patients: preliminary data. Clin Exp Rheumatol. 2015;33(1 Suppl 88):S82-85.
66. Rossini M et al. Double-blind, multicenter trial comparing acetyl l-carnitine with placebo in the treatment of fibromyalgia patients. Clin Exp Rheumatol. 2007;25(2):182-188.
67. Tardy AL et al. Vitamins and Minerals for Energy, Fatigue and Cognition: A Narrative Review of the Biochemical and Clinical Evidence. Nutrients. 2020;12(1).
68. Mikkelsen K et al. The Effects of Vitamin B in Depression. Curr Med Chem. 2016;23(38):4317-4337.
69. Zaric BL et al. Homocysteine and Hyperhomocysteinaemia. Curr Med Chem. 2019;26(16):2948-2961.
70. Young LM et al. A Systematic Review and Meta-Analysis of B Vitamin Supplementation on Depressive Symptoms, Anxiety, and Stress: Effects on Healthy and 'At-Risk' Individuals. Nutrients. 2019;11(9).
71. Almeida OP et al. Systematic review and meta-analysis of randomized placebo-controlled trials of folate and vitamin B12 for depression. International psychogeriatrics. 2015;27(5):727-737.
72. Mech AW et al. Correlation of clinical response with homocysteine reduction during therapy with reduced B vitamins in patients with MDD who are positive for MTHFR C677T or A1298C polymorphism: a randomized, double-blind, placebo-controlled study. The Journal of clinical psychiatry. 2016;77(5):668-671.
73. Tsujita N et al. Effect of Tryptophan, Vitamin B(6), and Nicotinamide-Containing Supplement Loading between Meals on Mood and Autonomic Nervous System Activity in Young Adults with Subclinical Depression: A Randomized, Double-Blind, and Placebo-Controlled Study. J Nutr Sci Vitaminol (Tokyo). 2019;65(6):507-514.
74. Roberts E et al. Caveat emptor: Folate in unipolar depressive illness, a systematic review and meta-analysis. Journal of psychopharmacology (Oxford, England). 2018;32(4):377-384.
75. Martone G. Enhancement of recovery from mental illness with l-methylfolate supplementation. Perspect Psychiatr Care. 2018;54(2):331-334.
76. Syed EU et al. Vitamin B12 supplementation in treating major depressive disorder: a randomized controlled trial. Open Neurol J. 2013;7:44-48.
77. Ghaleiha A et al. Adjuvant thiamine improved standard treatment in patients with major depressive disorder: results from a randomized, double-blind, and placebo-controlled clinical trial. European archives of psychiatry and clinical neuroscience. 2016;266(8):695-702.
78. Klinge CM et al. Dehydroepiandrosterone Research: Past, Current, and Future. Vitamins and hormones. 2018;108:1-28.
79. Stárka L et al. Dehydroepiandrosterone: a neuroactive steroid. J Steroid Biochem Mol Biol. 2015;145:254-260.
80. Hu Q et al. Clinical significance of decreased protein expression of dehydroepiandrosterone sulfate in the development of depression: a meta-analysis. J Affect Disord. 2015;174:416-423.
81. Mocking RJ et al. DHEAS and cortisol/DHEAS-ratio in recurrent depression: State, or trait predicting 10-year recurrence? Psychoneuroendocrinology. 2015;59:91-101.
82. Bloch M et al. Dehydroepiandrosterone treatment of midlife dysthymia. Biological psychiatry. 1999;45(12):1533-1541.
83. Schmidt PJ et al. Dehydroepiandrosterone monotherapy in midlife-onset major and minor depression. Archives of general psychiatry. 2005;62(2):154-162.
84. Wolkowitz OM et al. Double-blind treatment of major depression with dehydroepiandrosterone. The American journal of psychiatry. 1999;156(4):646-649.
85. Strous RD et al. Dehydroepiandrosterone augmentation in the management of negative, depressive, and anxiety symptoms in schizophrenia. Arch Gen Psychiatry. 2003;60(2):133-141.
86. Rabkin JG et al. Placebo-controlled trial of dehydroepiandrosterone (DHEA) for treatment of nonmajor depression in patients with HIV/AIDS. The American journal of psychiatry. 2006;163(1):59-66.
87. Jones LA et al. The ever-changing roles of serotonin. Int J Biochem Cell Biol. 2020:105776.
88. Kałużna-Czaplińska J et al. How important is tryptophan in human health? Crit Rev Food Sci Nutr. 2019;59(1):72-88.
89. Friedman M. Analysis, Nutrition, and Health Benefits of Tryptophan. Int J Tryptophan Res. 2018;11:1178646918802282.
90. Savitz J. Role of Kynurenine Metabolism Pathway Activation in Major Depressive Disorders. Curr Top Behav Neurosci. 2017;31:249-267.
91. Strasser B et al. Mood, food, and cognition: role of tryptophan and serotonin. Current opinion in clinical nutrition and metabolic care. 2016;19(1):55-61.
92. Ramírez LA et al. A new theory of depression based on the serotonin/kynurenine relationship and the hypothalamicpituitary- adrenal axis. Biomedica. 2018;38(3):437-450.
93. Dell'Osso L et al. Depression, Serotonin and Tryptophan. Curr Pharm Des. 2016;22(8):949-954.
94. Ramos-Chávez LA et al. Low Serum Tryptophan Levels as an Indicator of Global Cognitive Performance in Nondemented Women over 50 Years of Age. Oxid Med Cell Longev. 2018;2018:8604718.
95. Ogawa S et al. Plasma L-tryptophan concentration in major depressive disorder: new data and meta-analysis. The Journal of clinical psychiatry. 2014;75(9):e906-915.
96. Lieberman HR et al. Tryptophan Intake in the US Adult Population Is Not Related to Liver or Kidney Function but Is Associated with Depression and Sleep Outcomes. J Nutr. 2016;146(12):2609s-2615s.
97. Lindseth G et al. The effects of dietary tryptophan on affective disorders. Arch Psychiatr Nurs. 2015;29(2):102-107.
98. Kikuchi AM et al. A systematic review of the effect of L-tryptophan supplementation on mood and emotional functioning. J Diet Suppl. 2020:1-18.
99. Young SN. The effect of raising and lowering tryptophan levels on human mood and social behaviour. Philosophical transactions of the Royal Society of London Series B, Biological sciences. 2013;368(1615):20110375.
100. Gibson EL. Tryptophan supplementation and serotonin function: genetic variations in behavioural effects. The Proceedings of the Nutrition Society. 2018;77(2):174-188.
101. Jangid P et al. Comparative study of efficacy of l-5-hydroxytryptophan and fluoxetine in patients presenting with first depressive episode. Asian J Psychiatr. 2013;6(1):29-34.
102. Birdsall TC. 5-Hydroxytryptophan: a clinically-effective serotonin precursor. Altern Med Rev. 1998;3(4):271-280.
103. Jacobsen JPR et al. Adjunctive 5-Hydroxytryptophan Slow-Release for Treatment-Resistant Depression: Clinical and Preclinical Rationale. Trends Pharmacol Sci. 2016;37(11):933-944.
104. Das YT et al. Safety of 5-hydroxy-L-tryptophan. Toxicol Lett. 2004;150(1):111-122.
105. Deepmala et al. Clinical trials of N-acetylcysteine in psychiatry and neurology: A systematic review. Neuroscience and biobehavioral reviews. 2015;55:294-321.
106. Berk M et al. The efficacy of adjunctive N-acetylcysteine in major depressive disorder: a double-blind, randomized, placebo-controlled trial. The Journal of clinical psychiatry. 2014;75(6):628-636.
107. Porcu M et al. Effects of adjunctive N-acetylcysteine on depressive symptoms: Modulation by baseline high-sensitivity C-reactive protein. Psychiatry research. 2018;263:268-274.
108. Cullen KR et al. N-Acetylcysteine for Nonsuicidal Self-Injurious Behavior in Adolescents: An Open-Label Pilot Study. J Child Adolesc Psychopharmacol. 2018;28(2):136-144.
109. Fernandes BS et al. N-Acetylcysteine in depressive symptoms and functionality: a systematic review and meta-analysis. J Clin Psychiatry. 2016;77(4):e457-466.
110. Bottiglieri T. Folate, vitamin B₁₂, and S-adenosylmethionine. Psychiatr Clin North Am. 2013 Mar;36(1):1-13.
111. Sharma A et al. S-Adenosylmethionine (SAMe) for Neuropsychiatric Disorders: A Clinician-Oriented Review of Research. J Clin Psychiatry. 2017;78(6):e656-e667.
112. Williams AL et al. S-adenosylmethionine (SAMe) as treatment for depression: a systematic review. Clinical and investigative medicine Medecine clinique et experimentale. 2005;28(3):132-139.
113. Bressa GM. S-adenosyl-l-methionine (SAMe) as antidepressant: meta-analysis of clinical studies. Acta Neurol Scand Suppl. 1994;154:7-14.
114. Hardy ML et al. S-adenosyl-L-methionine for treatment of depression, osteoarthritis, and liver disease. Evid Rep Technol Assess (Summ). 2003(64):1-3.
115. Sarris J et al. S-Adenosylmethionine (SAMe) monotherapy for depression: an 8-week double-blind, randomised, controlled trial. Psychopharmacology. 2020;237(1):209-218.
116. Sarris J et al. Adjunctive S-adenosylmethionine (SAMe) in treating non-remittent major depressive disorder: An 8-week double-blind, randomized, controlled trial<sup/>. Eur Neuropsychopharmacol. 2018;28(10):1126-1136.
117. Galizia I et al. S-adenosyl methionine (SAMe) for depression in adults. The Cochrane database of systematic reviews. 2016;10(10):Cd011286.
118. Mischoulon D et al. A double-blind, randomized, placebo-controlled clinical trial of S-adenosyl-L-methionine (SAMe) versus escitalopram in major depressive disorder. The Journal of clinical psychiatry. 2014;75(4):370-376.
119. Sarris J et al. Is S-Adenosyl Methionine (SAMe) for Depression Only Effective in Males? A Re-Analysis of Data from a Randomized Clinical Trial. Pharmacopsychiatry. 2015;48(4-5):141-144.
120. Karas Kuželički N. S-Adenosyl Methionine in the Therapy of Depression and Other Psychiatric Disorders. Drug Dev Res. 2016;77(7):346-356.
121. Kious BM et al. Creatine for the Treatment of Depression. Biomolecules. 2019;9(9).
122. Bakian AV et al. Dietary creatine intake and depression risk among U.S. adults. Translational psychiatry. 2020;10(1):52.
123. Lyoo IK et al. A randomized, double-blind placebo-controlled trial of oral creatine monohydrate augmentation for enhanced response to a selective serotonin reuptake inhibitor in women with major depressive disorder. The American journal of psychiatry. 2012;169(9):937-945.
124. Yoon S et al. Effects of Creatine Monohydrate Augmentation on Brain Metabolic and Network Outcome Measures in Women With Major Depressive Disorder. Biological psychiatry. 2016;80(6):439-447.
125. Kious BM et al. An Open-Label Pilot Study of Combined Augmentation With Creatine Monohydrate and 5-Hydroxytryptophan for Selective Serotonin Reuptake Inhibitor- or Serotonin-Norepinephrine Reuptake Inhibitor-Resistant Depression in Adult Women. Journal of clinical psychopharmacology. 2017;37(5):578-583.
126. Kondo DG et al. Open-label adjunctive creatine for female adolescents with SSRI-resistant major depressive disorder: a 31-phosphorus magnetic resonance spectroscopy study. J Affect Disord. 2011;135(1-3):354-361.
127. Nemets B et al. A pilot dose-finding clinical trial of creatine monohydrate augmentation to SSRIs/SNRIs/NASA antidepressant treatment in major depression. International clinical psychopharmacology. 2013;28(3):127-133.
128. Colodny L et al. Inositol--clinical applications for exogenous use. Alternative medicine review : a journal of clinical therapeutic. 1998;3(6):432-447.
129. Zheng H et al. High-frequency rTMS treatment increases left prefrontal myo-inositol in young patients with treatment-resistant depression. Progress in neuro-psychopharmacology & biological psychiatry. 2010;34(7):1189-1195.
130. Moore CM et al. Effects of myo-inositol ingestion on human brain myo-inositol levels: a proton magnetic resonance spectroscopic imaging study. Biological psychiatry. 1999;45(9):1197-1202.
131. Levine J et al. Double-blind, controlled trial of inositol treatment of depression. The American journal of psychiatry. 1995;152(5):792-794.
132. Mukai T, Kishi T, Matsuda Y, Iwata N. A meta-analysis of inositol for depression and anxiety disorders. Human psychopharmacology. 2014;29(1):55-63.
133. Levine J et al. Combination of inositol and serotonin reuptake inhibitors in the treatment of depression. Biological psychiatry. 1999;45(3):270-273.
134. Nemets B et al. Inositol addition does not improve depression in SSRI treatment failures. Journal of neural transmission (Vienna, Austria : 1996). 1999;106(7-8):795-798.
135. Botturi A et al. The Role and the Effect of Magnesium in Mental Disorders: A Systematic Review. Nutrients. 2020;12(6).
136. Tarleton EK et al. The Association between Serum Magnesium Levels and Depression in an Adult Primary Care Population. Nutrients. 2019;11(7).
137. Sun C et al. Dietary magnesium intake and risk of depression. J Affect Disord. 2019;246:627-632.
138. Phelan D et al. Magnesium and mood disorders: systematic review and meta-analysis. BJPsych Open. 2018;4(4):167-179.
139. Tarleton EK et al. Role of magnesium supplementation in the treatment of depression: A randomized clinical trial. PLoS One. 2017;12(6):e0180067.
140. Rajizadeh A et al. Effect of magnesium supplementation on depression status in depressed patients with magnesium deficiency: A randomized, double-blind, placebo-controlled trial. Nutrition (Burbank, Los Angeles County, Calif). 2017;35:56-60.
141. Vink R. Magnesium in the CNS: recent advances and developments. Magnesium research. 2016;29(3):95-101.
142. Sun Q et al. Regulation of structural and functional synapse density by L-threonate through modulation of intraneuronal magnesium concentration. Neuropharmacology. 2016;108:426-439.
143. Kim YS et al. Neuroprotective effects of magnesium L-threonate in a hypoxic zebrafish model. BMC neuroscience. 2020;21(1):29.
144. Shen Y et al. Treatment Of Magnesium-L-Threonate Elevates The Magnesium Level In The Cerebrospinal Fluid And Attenuates Motor Deficits And Dopamine Neuron Loss In A Mouse Model Of Parkinson's disease. Neuropsychiatr Dis Treat. 2019;15:3143-3153.
145. Jia S et al. Elevation of Brain Magnesium Potentiates Neural Stem Cell Proliferation in the Hippocampus of Young and Aged Mice. J Cell Physiol. 2016;231(9):1903-1912.
146. Sadir S et al. Neurobehavioral and biochemical effects of magnesium chloride (MgCl2), magnesium sulphate (MgSO4) and magnesium-L-threonate (MgT) supplementation in rats: A dose dependent comparative study. Pakistan journal of pharmaceutical sciences. 2019;32(1(Supplementary)):277-283.
147. Ochoa-de la Paz L et al. Taurine and GABA neurotransmitter receptors, a relationship with therapeutic potential? Expert Rev Neurother. 2019;19(4):289-291.
148. Cui T et al. Abnormal levels of seven amino neurotransmitters in depressed rat brain and determination by HPLC-FLD. Biomed Chromatogr. 2017;31(8).
149. Caletti G et al. Antidepressant effect of taurine in diabetic rats. Amino Acids. 2012;43(4):1525-1533.
150. Caletti G et al. Antidepressant dose of taurine increases mRNA expression of GABAA receptor α2 subunit and BDNF in the hippocampus of diabetic rats. Behavioural brain research. 2015;283:11-15.
151. Wu GF et al. Antidepressant effect of taurine in chronic unpredictable mild stress-induced depressive rats. Sci Rep. 2017;7(1):4989.
152. O'Donnell CP et al. Adjunctive Taurine in First-Episode Psychosis: A Phase 2, Double-Blind, Randomized, Placebo-Controlled Study. The Journal of clinical psychiatry. 2016;77(12):e1610-e1617.
153. Asher GN et al. Comparative Benefits and Harms of Complementary and Alternative Medicine Therapies for Initial Treatment of Major Depressive Disorder: Systematic Review and Meta-Analysis. Journal of alternative and complementary medicine (New York, NY). 2017;23(12):907-919.
154. Kasper S et al. Better tolerability of St. John's wort extract WS 5570 compared to treatment with SSRIs: a reanalysis of data from controlled clinical trials in acute major depression. Int Clin Psychopharmacol. 2010;25(4):204-213.
155. Freeman MP et al. Complementary and alternative medicine for major depressive disorder: a meta-analysis of patient characteristics, placebo-response rates, and treatment outcomes relative to standard antidepressants. The Journal of clinical psychiatry. 2010;71(6):682-688.
156. Apaydin EA et al. A systematic review of St. John's wort for major depressive disorder. Syst Rev. 2016;5(1):148.
157. Herraiz T et al. Monoamine Oxidase-A Inhibition and Associated Antioxidant Activity in Plant Extracts with Potential Antidepressant Actions. Biomed Res Int. 2018;2018:4810394.
158. Sevastre-Berghian AC et al. Characterization and biological effects of Hypericum extracts on experimentally-induced - anxiety, oxidative stress and inflammation in rats. Journal of physiology and pharmacology : an official journal of the Polish Physiological Society. 2018;69(5).
159. Schmidt M et al. The mechanisms of action of St. John's wort: an update. Wien Med Wochenschr. 2015;165(11-12):229-235.
160. Nicolussi S et al. Clinical relevance of St. John's wort drug interactions revisited. Br J Pharmacol. 2020;177(6):1212-1226.
161. Blumenthal M et al. Herbal Medicine Expanded Commission E Monographs. Dallas, TX: American Botanical Council; 2000.
162. Khazdair MR at al. The effects of Crocus sativus (saffron) and its constituents on nervous system: A review. Avicenna J Phytomed. 2015;5(5):376-391.
163. Dai L et al. Safety and Efficacy of Saffron (Crocus sativus L.) for Treating Mild to Moderate Depression: A Systematic Review and Meta-analysis. The Journal of nervous and mental disease. 2020.
164. Khaksarian M et al. The efficacy of Crocus sativus (Saffron) versus placebo and Fluoxetine in treating depression: a systematic review and meta-analysis. Psychology research and behavior management. 2019;12:297-305.
165. Tóth B et al. The Efficacy of Saffron in the Treatment of Mild to Moderate Depression: A Meta-analysis. Planta Med. 2019;85(1):24-31.
166. Yang X et al. Comparative efficacy and safety of Crocus sativus L. for treating mild to moderate major depressive disorder in adults: a meta-analysis of randomized controlled trials. Neuropsychiatr Dis Treat. 2018;14:1297-1305.
167. Ghaderi A et al. The effects of saffron (Crocus sativus L.) on mental health parameters and C-reactive protein: A meta-analysis of randomized clinical trials. Complement Ther Med. 2020;48:102250.
168. Talaei A et al. Crocin, the main active saffron constituent, as an adjunctive treatment in major depressive disorder: a randomized, double-blind, placebo-controlled, pilot clinical trial. J Affect Disord. 2015;174:51-56.
169. Milajerdi A et al. The effects of alcoholic extract of saffron (Crocus satious L.) on mild to moderate comorbid depression-anxiety, sleep quality, and life satisfaction in type 2 diabetes mellitus: A double-blind, randomized and placebo-controlled clinical trial. Complement Ther Med. 2018;41:196-202.
170. Lopresti AL et al. Efficacy of a standardised saffron extract (affron®) as an add-on to antidepressant medication for the treatment of persistent depressive symptoms in adults: A randomised, double-blind, placebo-controlled study. Journal of psychopharmacology (Oxford, England). 2019;33(11):1415-1427.
171. Akhondzadeh S et al. A placebo controlled randomized clinical trial of Crocus sativus L. (saffron) on depression and food craving among overweight women with mild to moderate depression. J Clin Pharm Ther. 2020;45(1):134-143.
172. Kashani L et al. Efficacy of Crocus sativus (saffron) in treatment of major depressive disorder associated with post-menopausal hot flashes: a double-blind, randomized, placebo-controlled trial. Arch Gynecol Obstet. 2018;297(3):717-724.
173. Jalali F et al. The Effect of Saffron on Depression among Recovered Consumers of Methamphetamine Living with HIV/AIDS. Subst Use Misuse. 2018;53(12):1951-1957.
174. Shahmansouri N et al. A randomized, double-blind, clinical trial comparing the efficacy and safety of Crocus sativus L. with fluoxetine for improving mild to moderate depression in post percutaneous coronary intervention patients. J Affect Disord. 2014;155:216-222.
175. Shakiba M et al. Saffron (Crocus sativus) versus duloxetine for treatment of patients with fibromyalgia: A randomized double-blind clinical trial. Avicenna journal of phytomedicine. 2018;8(6):513-523.
176. Kashani L et al. Saffron for treatment of fluoxetine-induced sexual dysfunction in women: randomized double-blind placebo-controlled study. Human psychopharmacology. 2013;28(1):54-60.
177. Modabbernia A et al. Effect of saffron on fluoxetine-induced sexual impairment in men: randomized double-blind placebo-controlled trial. Psychopharmacology (Berl). 2012;223(4):381-388.
178. Kim M et al. Effects of Lavender on Anxiety, Depression, and Physiological Parameters: Systematic Review and Meta-Analysis. Asian Nurs Res (Korean Soc Nurs Sci). 2021 Dec;15(5):279-290.
179. Bazrafshan MR et al. The effect of lavender herbal tea on the anxiety and depression of the elderly: A randomized clinical trial. Complementary therapies in medicine. 2020;50:102393.
180. Kasper S et al. Efficacy of Silexan in mixed anxiety-depression--A randomized, placebo-controlled trial. Eur Neuropsychopharmacol. 2016;26(2):331-340.
181. Uehleke B et al. Phase II trial on the effects of Silexan in patients with neurasthenia, post-traumatic stress disorder or somatization disorder. Phytomedicine. 2012;19(8-9):665-671.
182. Akhondzadeh S et al. Comparison of Lavandula angustifolia Mill. tincture and imipramine in the treatment of mild to moderate depression: a double-blind, randomized trial. Progress in neuro-psychopharmacology & biological psychiatry. 2003;27(1):123-127.
183. Ross SM. Rhodiola rosea (SHR-5), Part 2: A standardized extract of Rhodiola rosea is shown to be effective in the treatment of mild to moderate depression. Holistic nursing practice. 2014;28(3):217-221.
184. Limanaqi F et al. Potential Antidepressant Effects of Scutellaria baicalensis, Hericium erinaceus and Rhodiola rosea. Antioxidants (Basel, Switzerland). 2020;9(3).
185. Concerto C et al. Exploring the effect of adaptogenic Rhodiola Rosea extract on neuroplasticity in humans. Complement Ther Med. 2018;41:141-146.
186. Mao JJ et al. Rhodiola rosea versus sertraline for major depressive disorder: A randomized placebo-controlled trial. Phytomedicine. 2015;22(3):394-399.
187. Darbinyan V et al. Clinical trial of Rhodiola rosea L. extract SHR-5 in the treatment of mild to moderate depression. Nord J Psychiatry. 2007;61(5):343-348.
188. Gao L et al. Antidepressants effects of Rhodiola capsule combined with sertraline for major depressive disorder: A randomized double-blind placebo-controlled clinical trial. J Affect Disord. 2020;265:99-103.
189. Amsterdam JD et al. Rhodiola rosea L. as a putative botanical antidepressant. Phytomedicine. 2016;23(7):770-783.
190. Araj-Khodaei M et al. A double-blind, randomized pilot study for comparison of Melissa officinalis L. and Lavandula angustifolia Mill. with Fluoxetine for the treatment of depression. BMC Complement Med Ther. 2020;20(1):207-207.
191. López V et al. Neuroprotective and neurological properties of Melissa officinalis. Neurochem Res. 2009;34(11):1955-1961.
192. Kirsch I. Placebo Effect in the Treatment of Depression and Anxiety. Frontiers in psychiatry. 2019;10:407.
193. Haybar H et al. The effects of Melissa officinalis supplementation on depression, anxiety, stress, and sleep disorder in patients with chronic stable angina. Clinical nutrition ESPEN. 2018;26:47-52.
194. Ranjbar M et al. Effects of Herbal combination (Melissa officinalis L. and Nepeta menthoides Boiss. & Buhse) on insomnia severity, anxiety and depression in insomniacs: Randomized placebo controlled trial. Integr Med Res. 2018;7(4):328-332.
www.mayoclinic.org
美国抑郁和双相障碍支持联盟
http://www.dbsalliance.org
美国国家公众健康网
www.medlienplus.gov
美国心理健康网
http://www.mentalhealthamerica.net
加拿大心理健康协会
http://www.ontario.cmha.ca
加拿大精神病协会
http://www.cpa-apc.org
免责声明和安全信息
英文名称:Depression
定义
抑郁症又称抑郁障碍,以显著而持久的心境低落、忧伤和对活动缺乏兴趣等为主要特征。情绪消沉可以从闷闷不乐到悲痛欲绝,或自卑抑郁,甚至悲观厌世、有自杀企图或行为,干扰了患者日常生活功能。每次发作持续至少2周以上,长者甚或数年,多数病例有反复发作的倾向。如果不治疗,抑郁症患者罕有能恢复。抑郁症一般包含以下几种主要类型:
- 重性抑郁(Major Depression):具有抑郁症的全部症状,且程度较重,有的发作少,也有反复发生的。
- 恶劣心境障碍(持续性抑郁障碍,或慢性轻度抑郁症):症状轻度到中度,也可能经历过重性抑郁。
- 非典型抑郁症:没有典型症状的抑郁症,可暂时为快乐事件高兴,食欲大增、睡眠过头,且常伴有躯体和精神多种不适,如头痛头晕、心悸、胸闷气短、四肢麻木等。
- 产后抑郁症:发生率约在10-15%,但往往被忽视。
- 双相情感障碍:也称狂躁抑郁症,表现为情绪低落和高涨两个极端。
- 季节性情感障碍:在中、高纬度地区的冬季发生和存在,与光照不足、引起体内维生素D合成不足有关。
- 精神病性抑郁症:严重抑郁症,有精神病特征,如幻觉、妄想,躲避现实世界。
病因
抑郁症的确切原因尚不清楚,但生物、心理和社会环境等因素发挥了重要作用。研究表明,抑郁与代谢因素相关,如氧化应激、炎症水平和胰岛素抵抗等,甚至线粒体障碍也参与其中,这些因素的存在及变化均可影响到脑化学失衡。与焦虑、抑郁等神经症相关的一些病因病理研究机制:
- 焦虑、抑郁障碍和下丘脑-垂体-肾上腺(HPA)轴:焦虑与抑郁大多由慢性应激引起,后者长期激活HPA,导致脑神经受损、神经元功能障碍及致脑结构改变等。
- 激素失衡与心境障碍:研究表明,大多数类固醇激素如孕烯醇酮、雌激素、孕酮、睾酮和DHEA具有神经活性,因为大脑中大量存在这些激素的受体。因此,荷尔蒙水平与焦虑、抑郁等情绪障碍密切相关。如女性月经周期、妊娠与产后、更年期和绝经后等,都可产生明显变化的神经症或情绪紊乱。此外,应激激素(即压力激素,糖皮质激素)也对抑郁症状存在明显的作用。
- 脑化学失衡: 甲基化依赖于B族维生素,尤其是B6,B12(甲钴胺)和叶酸,对于情绪调节中的神经递质合成至关重要;参与甲基化循环的另一种化合物是S-腺苷甲硫氨酸(SAMe),其提供甲基参与甲基化循环中,促进神经递质如多巴胺和血清素(5-羟色胺)的合成。
风险因素
抑郁症在女性中更易发生。有系列的可能增加患抑郁症的风险因素,包括如下:
- 抑郁症家族史
- 配偶抑郁
- 以前抑郁症发作
- 荷尔蒙的变化
- 多种慢性疾病,尤其精神类疾病
- 营养不良或失衡,如维生素B族、维生素D和多不饱和脂肪酸(鱼油和GLA)等
- 其他与心理健康有关的条件,如焦虑,吸毒,失眠和人格障碍
- 颅脑外伤史
- 青少年脑震荡的历史
- 药物或酒精使用障碍的历史
- 家庭暴力
- 儿童遭受虐待、欺负、忽视或性虐待史
- 青少年自残历史,有或无自杀意图
- 情感或社会因素,如:
- 很少或根本没有社会支持互动
- 消极的思维模式和信念
- 低自尊
- 对环境缺乏控制
- 无助的感受
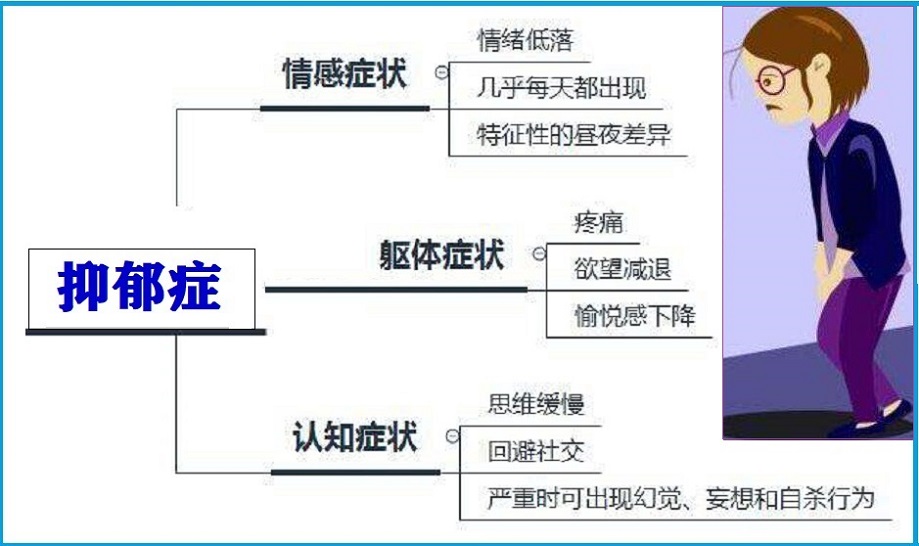
症状
抑郁症状可因人而异。有些人只有很少的症状,而其他人却有许多,而且症状会随着时间而改变。症状一般包括如下:- 精神情绪症状如:
- 持续的或短暂的悲伤、哭泣、焦虑和空虚
- 感觉躁动、易怒
- 感到无望、绝望
- 小事情也生气、爆发脾气
- 毫无理由的内疚、负罪或自责,甚至有自杀念头
- 入睡困难、早醒,或睡眠少,或太多
- 身体症状可包括如下
- 疲乏、无力
- 体重增加或减少,因为吃比往常多或少
- 注意力难以集中,难记忆细节,或做决定
- 丧失爱好和活动的兴趣
- 对性生活失去兴趣
- 躯体症状无法诊断和对治疗反应不良
并发症
抑郁症是一种严重的疾病,可给患者及其家人带来可怕的伤害。如果不控制好,抑郁症往往会变得更糟,导致情绪、行为和健康等多方面问题。有关的并发症包括:
- 体重过重或肥胖,可导致心脏病和糖尿病
- 疼痛或躯体疾病
- 酒精或药物滥用
- 焦虑、恐慌症或社交恐惧症
- 家庭冲突
- 社交隔离,或社会关系紧张
- 自残,如切割
- 自杀企图或自杀
- 由于医疗条件而过早死亡
疗法
严重的抑郁症患者,可能需要住院治疗。综合治疗更有效,选项可包括如下:
调整饮食和生活方式
改变饮食包括如下:
- 饮食富含不饱和脂肪酸食物,如深海鱼、植物坚果种子、亚麻籽等
- 健康饮食,包括水果、蔬菜及全谷类食物
- 限制刺激性食物,如酒精、咖啡因等
- 经常运动锻炼,对抑郁症控制有效
- 获得充足的睡眠和休息
- 积极参与社交和各种娱乐
营养与草本综合干预
以下是基于循证医学和循证营养学有关文献综合的结果。
有助于防控抑郁症的营养和草本补充剂,主要包括如下:
1.欧米伽3脂肪酸:
EPA和DHA是神经细胞膜的重要组成部分,影响其流动性和信号传导能力1。它们也在体内被用来产生抗炎代谢物,以及内源性大麻素,一种通过与大麻素受体相互作用来调节神经功能的化合物。这些效果与重度抑郁症的改善有关2,3。临床前研究进一步表明,EPA和DHA可增强神经可塑性4。
适量食用富含脂肪的深海鱼类(EPA、DHA主要膳食来源),可以降低患抑郁症的风险3,5。在患有抑郁症和其他精神疾病的人中,较高水平的ω-3脂肪酸与较轻的症状相关6,7。
大量随机对照试验发现,以2:1或3:1的比例服用EPA和DHA补充剂,每天服用1-2克,对抑郁症有积极影响8,9。欧米伽3脂肪酸也可能对抑郁症和代谢紊乱(如2型糖尿病、肥胖症和心血管疾病)患者有代谢益处10-13。基于大量证据,国际营养精神病学研究学会((ISRPN))建议在治疗和预防严重抑郁症时使用EPA14。还应认识到DHA在支持情绪健康方面的独特作用。DHA是中枢神经系统中最常见的脂肪酸。DHA不仅被整合到神经细胞膜中,这是正常结构和功能所必需的,它也是其他脑细胞的关键组成部分,这些脑细胞利用DHA来支持神经元生长和神经连接的形成。此外,DHA是一种关键的抗炎物质的来源(称为Resolvins和Protectins)3,15。
2. 锌:
锌在体内具有多种功能,包括减少氧化应激、调节神经递质受体功能和调节基因表达16,17。重要的是,锌可以抑制神经元NMDA谷氨酸受体,而NMDA谷氨酸在抑郁症患者中失调,并提高BDNF水平,可促进神经可塑性18,19。对九项研究的荟萃分析发现,低锌摄入与抑郁症风险增加有关20。330此外,据报道,与非抑郁症患者相比,抑郁症患者的锌水平较低,锌缺乏与抑郁症的严重程度和治疗耐药性的增加有关18,21。已经发现,补充锌作为一种独立的治疗方法,与抗抑郁治疗相结合,即使在治疗耐药性抑郁症的情况下,也能改善抑郁症19,22。
一项针对50名超重和肥胖受试者的对照试验发现,抑郁症症状与较低的血液BDNF水平相关,连续12周每天补充30mg锌比安慰剂更有效地提高了BDNF水平并减轻了抑郁症症状23。在30名健康年轻女性中,与单独服用多种维生素相比,在10周内每天补充7毫克锌会降低评估愤怒和敌意的分数24。一项为期12周的试验包括44名患有严重抑郁症的参与者,发现那些除了服用常规的SSRI外,还服用硫酸锌(每天提供25mg元素锌)的人与那些服用SSRI的人相比,抑郁症的改善更大25。在一项对14名重度抑郁症患者进行SSRIs或三环类抗抑郁药治疗的试验中,发现相同剂量的锌在降低抑郁评分方面比安慰剂更有益26。在60例难治性抑郁症患者中,与安慰剂相比,在三环抗抑郁药丙咪嗪(Tofranil)治疗12周后,每天添加25mg锌可获得更快速、更有力的治疗反应27。
3.维生素D:
许多观察性研究注意到低维生素D水平与抑郁症风险之间的联系28-30,一些研究注意到维生素D缺乏与自杀之间的联系31,32。此外,维生素D水平低也可能与抑郁症患者的认知障碍有关33。维生素D与抑郁症之间的联系可能与维生素D的免疫调节抗炎作用、促进神经可塑性和神经发生的能力及其对神经递质水平的影响有关30,34。
一项随机安慰剂对照试验对18,353名50岁及以上的参与者(其中16,657人从未患过抑郁症,1696人患有抑郁症,但至少两年未接受治疗)进行了研究,结果发现在平均5.3年的时间内每天服用2000IU维生素D并没有降低患抑郁症或临床相关抑郁症状的风险35。然而,这项研究的方法和严谨性受到了质疑。305在多项临床研究中,发现维生素D补充剂对抑郁症的有益作用。在临床试验中发现,维生素D可以有效地减轻抑郁症患者的症状,每天服用2800至5600IU,持续1至12个月30。一项对948名重度抑郁症患者进行的四项随机对照试验的荟萃分析发现,补充维生素D对抑郁症症状有实质性的积极影响36。另一项包括9项研究的荟萃分析发现,补充维生素D可以减轻抑郁症症状,改善各种精神病诊断患者的炎症和氧化应激的一些标志物37。
维生素D对健康有更广泛的益处,并可能减轻其他慢性病患者的抑郁情绪。在一项试验中,与12周后接受安慰剂治疗的受试者相比,每天接受4000IU维生素D治疗的2型糖尿病和轻度至中度抑郁症状受试者在情绪和代谢健康指标方面有更大的改善38。一项开放标签试验发现,每天10,000 IU维生素D,持续12个月,可减轻35名多发性硬化症患者的抑郁症状。血液测试显示,研究对象的维生素D水平在研究开始时缺乏或接近缺乏,并在研究过程中变得充足。此外,在整个研究过程中,较高的维生素D水平与较低的抑郁测试分数相关39。
4.益生菌:
肠道微生物组与大脑有着密切的关系,在调节情绪和认知功能方面发挥着重要作用。研究发现,抑郁症患者肠道微生物丰度和多样性降低,这种模式通常与健康状况较差有关40。在一项研究中,食用富含益生菌的发酵食品与患严重抑郁症的风险较低有关41。
多项临床试验、综述和荟萃分析表明,益生菌补充剂可以有效治疗抑郁症42-46。在一项为期8周的试验中,110名患者接受了轻度至中度抑郁症的抗抑郁药物治疗,接受由瑞士乳杆菌R00052和长双歧杆菌R00175组成(Lallemand益生菌专有品牌CEREBIOME®)的100亿CFU的患者在抑郁症测试中的得分比接受安慰剂的患者降低得更多47。这些相同的益生菌菌株以前被证明可以减轻健康志愿者的心理困扰48。一项安慰剂对照试验90名轻度至中度抑郁症受试者发现,在六周内每天服用一种能提供10亿CFU植物乳杆菌补充剂,以及200mg S-腺苷蛋氨酸(SAMe),有效地降低了抑郁症症状评分以及与抑郁症相关的焦虑和认知变化49。在40名重度抑郁症患者中,每天补充20亿CFU的嗜酸乳杆菌、干酪乳杆菌和两歧双歧杆菌、持续8周,可减轻抑郁症状,改善胰岛素抵抗、氧化应激和炎症的一些指标,效果优于安慰剂50。
在一项对40名治疗抵抗性抑郁症患者进行的公开试验中,在标准抗抑郁治疗中每天添加60mg含有丁酸梭菌(Clostridium butyricum)的补充剂,有效率为70%,缓解率为35%51。另一项针对治疗抵抗性抑郁症患者的小型开放试验发现,在抗抑郁治疗中加入一种由嗜酸乳杆菌、两歧双歧杆菌和嗜热链球菌组成的混合益生菌,加上每天1600mg的乳清酸镁,8周后12名参与者中有8名(67%)的抑郁和生活质量评分得到改善52。
益生菌可以减轻其他慢性病患者的抑郁症状。一项包括54名患有2型糖尿病和冠心病的参与者的安慰剂对照试验显示,每天服用嗜酸乳杆菌、罗伊氏乳杆菌、发酵乳杆菌和两歧双歧杆菌各20亿CFU的混合益生菌,以及200 mcg硒,持续12周,可以降低抑郁评分,改善代谢标志物53。同样,一项针对44名心脏病发作后患者的安慰剂对照试验发现,在12周内,每天16亿CFU鼠李糖乳杆菌可以减轻抑郁,减少炎症和氧化应激标志物,提高生活质量54。其他临床试验的结果表明,益生菌可以减轻肠易激综合征患者的抑郁症状55,56。
在一项值得注意的临床试验中,对155名健康人和156名重度抑郁症患者进行了研究,以确定其肠道微生物组和粪便代谢的细菌景观和代谢特征的差异57。试验参与者在三种噬菌体(感染细菌的病毒)、50种粪便代谢物和47种细菌的丰度方面存在显著差异。细菌种类主要分为三个属(分类学类别)之一:拟杆菌属(Bacteroides)、蓝杆菌属(Blautia)和真杆菌属(Eubacterium)。这些属的细菌种类多样,但在体内发挥关键作用,包括调节炎症57,58。重度抑郁症组的拟杆菌丰度显著增加,蓝杆菌属和真杆菌属丰度显著降低。
基于这些发现,作者认为这些属中的某些物种可能是导致重度抑郁症患者常见的炎症失衡的原因。重要的是,这项研究有助于为使用肠道微生物组特征来评估重度抑郁症铺平道路。
5.乙酰L肉碱:
乙酰-L-肉碱是L-肉碱的一种形式,对正常的线粒体能量代谢很重要。乙酰-L-肉碱很容易穿过血脑屏障,增强神经元线粒体代谢59。它被认为具有与抗炎和抗氧化应激机制有关的大脑保护作用,并促进神经可塑性60,61。据报道,抑郁症受试者血液中的乙酰-L-肉碱水平低于非抑郁症受试,并且水平随着抑郁症发病年龄的降低和严重程度的增加而下降;难治性抑郁症和有儿童创伤史的患者的水平最低62。
一项对12项随机对照试验(共791名参与者)进行的荟萃分析发现,乙酰-L-卡尼汀比安慰剂更有效,在减少抑郁症状方面,其疗效与广泛处方的抗抑郁药物相似63。一项对80名患有阈下抑郁症的老年患者进行的为期7周的安慰剂对照试验发现,乙酰-L-肉碱在减轻症状方面与抗抑郁药氟西汀一样有效,但是,乙酰基-L-肉碱组的症状改善比氟西汀更快64。已经发现,每天1500mg的乙酰L肉碱比安慰剂更有效地减轻纤维肌痛综合征患者的抑郁症状和疼痛,并改善生活质量,疗效类似于度洛西汀(Cymbalta)65,66。
6. 维生素B族:
维生素B复合物由九种水溶性B族维生素组成,它们在各种生化反应中起着相关的辅助因子作用,是正常细胞代谢和能量生产所必需的67。正常神经功能需要维生素B1(硫胺素)、维生素B3(烟酸)、维生素B6(吡哆醇)、B9(叶酸)和维生素B12,它们的缺乏与抑郁症有关68。将同型半胱氨酸转化为S-腺苷蛋氨酸(SAMe)需要足够量的三种甲基化酶的B6、B9和B12,SAMe是一种参与合成许多关键生物分子(包括神经递质)的甲硫氨酸。这三种B族维生素中的任何一种缺乏都会提高同型半胱氨酸水平。高同型半胱氨酸水平与抑郁症、心血管、神经系统和其他慢性疾病的风险增加有关69。
临床试验表明,补充B族维生素可以降低健康人的压力水平,改善情绪70。长期补充叶酸和B12可以降低重度抑郁症患者复发的风险,并且可能有助于预防高危人群患上严重抑郁症71。在患有严重抑郁症和导致同型半胱氨酸代谢中断的基因突变的患者中,服用含有代谢形式的B族维生素的多种维生素矿物质补充剂可以降低同型半胱氨酸水平,在减轻抑郁症状方面比安慰剂更有效,8周后缓解率为42%72。在一项为期7天的试验中,发现每天在两餐之间服用200 mg色氨酸、8mg B6和8mg B3的组合比安慰剂更好地减轻症状,该试验包括30名患有严重抑郁症症状但未达到严重抑郁症诊断阈值的年轻人73。
尽管对抑郁症患者的研究结果喜忧参半,但安慰剂对照试验表明,叶酸,特别是甲基叶酸,可能改善抗抑郁治疗的反应74,75。补充维生素B12也被发现可以减轻服用抗抑郁药的患者的症状:在一项包括73名服用抗抑郁药物治疗中重度抑郁症的患者的试验中,在每周肌肉注射1000mcg B12并持续六周的参与者中,100%的人的抑郁评分有20%或更高的改善,而未接受B12注射的参与者中只有69%的人有同样程度的改善76。在一项针对51名严重抑郁症住院患者的安慰剂对照试验中,每天在抗抑郁治疗中添加300mg 维生素B1可以更快地减轻症状77。
7.脱氢表雄酮:
脱氢表雄酮(DHEA)是一种肾上腺激素,可用于产生睾酮和雌二醇。它还具有免疫调节和代谢作用,独立于其作为性激素前体的功能78。DHEA有助于情绪调节,具有神经保护作用,并具有抗应激活性79。抑郁症患者的DHEA-硫酸盐(DHEA-s)水平较低,皮质醇与DHEA-s的比值被认为是衡量抑郁症中应激激素活性的指标80。抑郁症缓解期间皮质醇与DHEA比值的持续紊乱可能是复发风险的指标81。
在一项随机对照试验中,包括17名中年发作的非重度抑郁症参与者,与安慰剂相比,连续三周每天服用90mg DHEA可显著改善抑郁症症状82。同样,一项安慰剂对照试验涉及46名患有严重或非严重中年抑郁症的参与者,该试验也注意到DHEA的抗抑郁作用83。在另一项试验中,22名患有严重抑郁症的参与者(其中一些人正在接受抗抑郁治疗)在六周内每天接受90mg DHEA或安慰剂治疗。与对照组相比,DHEA组中更多的参与者出现了治疗反应,即抑郁症状减少50%84。据报道,DHEA还可以减轻精神分裂症85和HIV/AIDS患者的抑郁症状86。
8. 色氨酸和5-羟色氨酸:
L-色氨酸是肠道和大脑中血清素的氨基酸前体,是肠道和中枢神经系统之间交流的重要介质73,87,88。L-色氨酸也是褪黑素、维生素B3以及其他几种代谢物的前体。色氨酸代谢的改变会增加炎症代谢物的产生,这可能是抑郁症风险的一个因素,并将抑郁症与其他慢性疾病联系起来89-91。一些证据表明,压力会破坏正常的色氨,酸代谢,干扰血清素的产生,导致炎症和抑郁风险增加92,93。L-色氨酸水平已被注意到随着年龄的增长而降低94。
一项对研究的荟萃分析发现,重度抑郁症患者的循环色氨酸水平较低95。色氨酸摄入增加与抑郁症状减轻和社交能力增强有关,随机对照试验表明,每天补充3g L-色氨酸可以改善健康成年人的积极情绪96-99。影响色氨酸代谢的遗传因素似乎在一定程度上决定了个体对L-色氨酸酯补充的反应性100。
5-羟色氨酸(5-HTP)是色氨酸代谢的副产物,是血清素的直接前体。补充5-色氨酸而不是L -色氨酸可以避免其他L-色氨酸代谢物的产生,并且可以绕过色氨酸转化为5-羟色胺的限速步骤。随机对照试验发现,每天服用200-300mg 5-HTP比安慰剂更有效,在治疗重度抑郁症方面可与一线抗抑郁药相当101,102。一些证据表明5-羟色胺还可能增加抗抑郁治疗的反应性。5-羟色胺可引起消化不良,但与血清素综合征或严重的不良反应无关103,104。
9. N-乙酰半胱氨酸:
已知N-乙酰半胱氨酸(NAC)是半胱氨酸的衍生物。作为半胱氨酸的来源,它有助于提升谷胱甘肽水平,谷胱甘肽是代谢毒素和减少全身氧化应激所必需的。NAC在大脑中具有重要功能,可减少氧化应激和神经炎症,增强线粒体活性,调节多巴胺和谷氨酸信号传导,促进神经可塑性等重要功能105。
一项针对252名重度抑郁症参与者的对照试验将标准护理加每日2000mg NAC与标准护理加安慰剂进行了比较。在抑郁症最严重的患者中,从第六周开始,NAC的使用使症状得到了统计学上的显著改善,并一直持续到研究结束。此外,在试验结束后四周的随访中,NAC使用者的功能损伤得到了更大的改善106。另一项试验发现,每天1800mg NAC可减轻双相或重度抑郁症患者的抑郁和焦虑症状,C反应蛋白(系统炎症的标志物)基线水平升高107。在一项对35名有非自杀性自伤史的女性青年的试验中,NAC的剂量逐渐增加到每天3600mg,可以减轻抑郁症状和自残行为108。多项临床试验发现,NAC可以减轻抑郁症和其他精神和神经疾病患者的抑郁症状109。
10. S-腺苷蛋氨酸:
S-腺苷甲硫氨酸(SAMe)是合成许多关键生物分子(包括神经递质和激素)所需的单碳甲基的重要来源。从甲硫氨酸(或称蛋氨酸)生产SAMe需要足够量的叶酸和维生素B12,它们在化学反应中起辅助因子的作用110。研究发现,抑郁症患者的SAMe水平降低111。对2005年的研究进行的系统综述以及2003年和1994年发表的两项荟萃分析都发现了支持SAMe抗抑郁作用的证据112-114;然而,最近的试验无法明确证实这些发现111。
在一项包括49名轻度至中度抑郁症患者的安慰剂对照试验中,每天800mg SAMe持续8周,可显著减轻抑郁症症状,尤其是轻度抑郁症患者;然而,由于研究参与者人数较少,安慰剂反应非常强烈,SAMe的改善在统计学上没有显著意义115。同样,在对单独药物反应不足的患者中,将SAMe添加到抗抑郁治疗中,可导致具有临床意义但无统计学意义的改善,因为安慰剂组的反应率为50%116。
2016年,一项对8项试验的系统综述共包括934名受试者,发现SAMe与某些抗抑郁疗法具有相似的疗效,但总体证据质量较差117。一项为期12周的随机对照试验,189名重度抑郁症患者每天服用1600–3200mg SAMe,或安慰剂组发现三者效果相同118;然而,对这些发现的第二次分析显示,SAMe在男性中优于安慰剂,但在女性中则不然119。性别的潜在影响可以部分解释多年来临床试验结果的不一致性。
总体而言,现有证据不足以得出SAMe治疗抑郁症的疗效结论。对服用抗抑郁药的人进行SAMe辅助治疗可能是有效的,但需要更高质量的研究来阐明益处的程度(如果有的话)111。SAMe是双相情感障碍患者的禁忌症,因为据报道它在某些情况下会诱发躁狂120。
11.肌酸:
肌酸存在于肉类和鱼类中。它在调节高活性细胞(如肌肉和脑细胞)的能量产生方面发挥着关键作用。饮食中肌酸摄入量低与抑郁症风险增加有关,大脑肌酸代谢的改变已被确定为抑郁症的潜在因素121,122。
一项针对52名重度抑郁症女性的随机对照试验发现,艾司西酞普兰加肌酸一水合物联合治疗,每天3g,持续一周,然后每天5g,持续七周,与艾司西酞普兰加安慰剂相比,治疗反应性更好123。一项后续研究发现,补充肌酸也会增加大脑中神经连接的形成124。在一项针对15名耐治疗抑郁症女性的公开试验中,在8周内每天添加5g肌酸一水合物加100mg 5-HTP可使抑郁症状评分降低60%125。一项针对5名对氟西汀无反应的重度抑郁症少女的公开试验发现,在8周内向每天添加4g肌酸可使症状评分降低56%126。
然而,一项针对18名三周后对抗抑郁治疗无反应的抑郁症女性的对照试验发现,每天添加5或10g肌酸在提高反应率方面并不比安慰剂更有效127。有必要对肌酸进行进一步的研究,以阐明肌酸是否比安慰剂对重度抑郁症患者有益。
12.肌醇:
肌醇存在于许多动植物食品中。肌醇以磷脂酰肌醇及其衍生物的形式储存在细胞膜中。在中枢神经系统中,肌醇有助于调节神经元信号:神经细胞上神经递质受体的激活刺激肌醇从细胞膜输送到细胞中,在细胞中通过神经递质释放调节信号的持续存在128。据报道,抑郁症患者的脑内肌醇水平较低,并且可能由于成功的治疗而正常化128,129。口服肌醇补充剂可能有助于使大脑肌醇水平正常化128,130。
一项针对28名抑郁症参与者的随机对照试验发现,在为期四周的时间里,每天12g肌醇比安慰剂更有效地减轻症状131。在几个对照试验中,肌醇也被发现比安慰剂略好地减轻抑郁症症状132。然而,研究人员未能发现,与安慰剂相比,在接受SSRI治疗失败的抑郁症患者中,肌醇的益处133,134。
13.镁:
镁在大脑中具有许多重要功能,包括参与神经信号传递的调节135。一些研究报告称,较高的镁摄入量和血清水平与较低的抑郁可能性和抑郁症状的减少有关136,137。一些非对照试验发现,补充镁对减少抑郁症状有效138。在一项包括126名患者的交叉试验中对于轻度至中度抑郁症,在六周内每天补充248mg元素镁(氯化镁)与不治疗相比,症状显著减轻139。尽管大多数安慰剂对照试验发现镁的治疗效果与安慰剂没有什么不同138,一项试验发现,在60名镁缺乏的抑郁症患者中,每天500mg氧化镁持续8周,比安慰剂显著减轻症状140。
人们认为,镁穿过血脑屏障的能力差可能是其在临床试验中表现不佳的原因141。在细胞和动物研究中,苏糖酸镁(一种更容易渗透到中枢神经系统的镁盐)被发现可以提高大脑镁水平,改善大脑结构和功能142-145,在一项针对大鼠的研究中,苏糖酸镁比其他形式的镁更能减轻抑郁样症状,改善记忆力146。
14.牛磺酸:
中枢神经系统是牛磺酸的高聚集区,牛磺酸可以减少大脑氧化应激,刺激神经生长和发育,并参与神经信号传递的调节147。在实验诱发的抑郁症动物的大脑中,牛磺酸、天冬氨酸、甘氨酸和γ氨基丁酸(GABA)的水平都有所下降148。一个研究小组在糖尿病实验动物中发现了牛磺酸的抗抑郁作用,并指出补充牛磺酸可以逆转糖尿病相关的GABA信号传导和BDNF产生的减少,防止脑萎缩并改善记忆149,150。据报道,牛磺酸还能通过调节应激激素水平和促进神经生长、神经存活和新神经的形成,减少暴露于慢性应激的动物的抑郁样症状151。
在一项随机对照试验中,86名因首次精神病发作而接受治疗的患者每天服用4g牛磺酸或安慰剂,同时接受标准药物治疗12周;接受牛磺酸治疗的患者在精神症状(包括抑郁症状)和整体功能方面有更大的改善152。
15.圣约翰草:
多项临床试验表明,圣约翰草(贯叶连翘)可以减轻重度抑郁症的症状,其疗效与一线抗抑郁药物相似,但副作用更少153-155。一项荟萃分析包括35项随机对照试验,共有6,993名参与者,证实了圣约翰草对轻度至中度抑郁症的益处。然而,人们对它在严重的抑郁症中的作用知之甚少156。
圣约翰草中的一些活性化合物对单胺氧化酶有微弱的抑制作用,单胺氧化酶是一种分解神经递质的酶,而其他成分具有抗炎和清除自由基的作用157,158。研究认为其抗抑郁特性与多种活性化合物的协同作用有关159。标准化提取物通常含有0.1-0.3%的金丝桃素(Hypericin)和高达6%的贯叶金丝桃素(Hyperforin)160。
圣约翰草的提取物,尤其是那些富含贯叶金丝桃素的提取物,可以促进肠壁和肝脏的解毒活动,这可能影响许多药物的代谢。因此,服用药物并想要添加圣约翰草的人使用低贯叶金丝桃素制剂是最安全的,并应仔细监测患者服用药物的条件160。
在欧洲,圣约翰草是最常用的抗抑郁药之一,在德国的处方量几乎是所有其他处方抗抑郁药的二倍161。
16.藏红花:
藏红花及其活性成分因其对抑郁症的影响而被广泛研究。其抗抑郁作用可能是由于其抗炎作用、提高多巴胺水平的能力以及与内源性阿片受体的相互作用162。多项对照临床试验和荟萃分析表明,藏红花提取物(通常每天30mg)比安慰剂更有效,在改善轻度至中度抑郁方面与抗抑郁药物相当163-166。一项荟萃分析包括21项对照试验的结果,发现藏红花提取物可以降低自我报告的抑郁和焦虑评分,改善睡眠质量167。
在一项针对40名重性抑郁症患者的安慰剂对照试验中,与单独的抗抑郁治疗相比,抗抑郁治疗加上每天30mg番红花苷(藏红花中的一种活性化合物)持续4周的联合治疗,在减轻症状方面更有效168。在54名2型糖尿病和轻度至中度合并抑郁和焦虑的患者中,与安慰剂相比,每天服用30mg藏红花提取物8周后,情绪症状和睡眠改善更多169。在139名尽管接受了抗抑郁药物治疗但仍患有持续性抑郁症的患者中,添加标准化藏红花提取物,每天28mg,8周后降低了医生评估的抑郁症评分,但没有自我评估评分,优于安慰剂170。
藏红花提取物已被证明可以降低超重女性171、有更年期症状的健康女性172、正在药物滥用康复中的HIV/AIDS患者173、冠状动脉疾病患者174、纤维肌痛综合征患者175的抑郁症状。安慰剂对照临床试验进一步表明,藏红花与抗抑郁药联合使用,可以减轻男性和女性与性功能障碍相关的抗抑郁药副作用176,177。
17.薰衣草:
初步研究表明,薰衣草芳香疗法可以缓解抑郁症状。在一些小型研究中,口服薰衣草制剂如提取物、茶或酊剂,已被证明可以改善抑郁症状178。在一项单盲临床试验中,60名老年受试者被分配每天喝两次薰衣草茶或两周内不接受治疗。情绪评分采用标准化问卷进行评估。与不喝任何薰衣草茶的人相比,薰衣草茶组的抑郁症得分显著下降179。
在一项招募318名患有混合焦虑和抑郁的成年人的试验中,薰衣草提取物(Silexan,薰衣草口服品牌Silexan™)每天80mg、持续治疗70天后,在标准化抑郁评分表上的得分显著高于安慰剂180。在一项单独的开放标签试验中,对报告焦虑和不安的人进行了相同提取物的研究,治疗6周后,抑郁得分比基线显著下降181。
在另一项试验中,45名患有严重抑郁症的成年人被随机分配接受薰衣草酊(50%酒精中的1:5提取物)加安慰剂、薰衣草酊加抗抑郁药丙咪嗪或丙咪嗪加安慰剂治疗四周。研究发现,薰衣草和丙咪嗪的组合在减轻抑郁方面比单独使用丙咪嗪更有效,但单独使用薰衣草的效果不如单独使用丙咪嗪。研究人员得出结论:“这项研究的主要总体发现是,作为辅助治疗,[薰衣草]酊剂可能对轻度至中度抑郁症有治疗益处。”182
18.红景天:
作为适应原,红景天是一种提高应激弹性的疗法,历史上一直被用于治疗疲劳、增强耐力和促进长寿183。临床前证据表明,红景天调节神经递质信号传导并使应激反应正常化。它还具有抗炎和氧化应激减轻作用,这可能有助于其抗抑郁活性,并已被证明在健康志愿者的大脑中抑制抑郁样信号传导184-186。
一项针对89名轻度至中度抑郁症患者的对照试验发现,与安慰剂相比,每天340mg和680mg的红景天提取物在42天后减轻了抑郁症症状。尽管两种剂量的总体效果相似,但注意到接受较高剂量的患者自尊显著改善,而接受较低剂量的患者则没有观察到这一点187。在一项随机对照试验中,100名轻度至中度抑郁症患者接受了SSRI舍曲林加上每天300mg的红景天的治疗,每天600mg的红景天或安慰剂。12周后,接受高剂量红景天治疗的患者症状改善最大188。此外,多项公开试验的结果进一步证明红景天具有有意义的抗抑郁活性189。在一项针对57名轻度至中度抑郁症患者的12周比较试验中,与安慰剂相比,红景天提取物的剂量增加(每天340–1360mg)导致改善的可能性略低于舍曲林,但不良副作用较少186。
19.香蜂草:
香蜂草(Melissa officinalis)在欧洲历来被用于治疗情绪障碍,包括抑郁症和焦虑症,以及管理压力和促进睡眠190。当代临床前研究表明,香蜂草具有一些药理学特性,这可能部分解释了它影响情绪的能力。香蜂草提取物已在体外显示出抑制单胺氧化酶A(MAO-A)的作用,这种作用可以调节参与情绪调节的单胺神经递质的代谢191。
在一项小型试点研究中,45名患有严重抑郁症的成年人被随机分配服用氟西汀、香蜂草或另一种草药英国薰衣草(Lavandula angustifolia)190。名受试者在基线、两周、四周和八周时使用HAM-D评估情绪。在研究过程中,香蜂草和薰衣草在降低受试者HAM-D评分方面与氟西汀一样有效。这项研究的作者承认了这项工作的初步性质:“尽管在我们的研究中,M.officinalis和L.angustifolia的效果与氟西汀相当,但一项荟萃分析显示,这些效果[与安慰剂效应192。关于这一争议,需要更大规模的随访时间更长的试验,包括安慰剂组,以评估这些草药的长期安全性和有效性。”
早期的一项随机安慰剂对照试验评估了香蜂草对80名慢性稳定型心绞痛患者情绪和睡眠障碍的影响。参与者被随机分配接受每天3g香蜂草或安慰剂治疗,为期8周。使用标准化问卷对情绪进行评估。在研究结束时,服用香蜂草的受试者在抑郁、焦虑、压力和睡眠障碍方面的得分与服用安慰剂的受试对象相比显著降低193。另一项小型试验发现,香蜂草有助于缓解失眠者的抑郁情绪。在这项研究中,香蜂草与另一种草药薄荷联合使用,经过四周的治疗,这种组合比安慰剂更能改善情绪相关评分194。
更多内容可点击其个性化综合干预方案如下:
- 抑郁症防控要略(抗抑郁情绪)
- 抑郁症防控要略(支持脑生化)
- 抑郁症防控要略(改善脑功能)
- 抑郁症防控要略(防止脑损伤)
- 抑郁症防控要略(改善药物疗效)
- 抑郁症防控要略(缓解疲乏)
- 抑郁症防控要略(抗代谢障碍)
- 抑郁症防控要略(防止复发)
其他抑郁症管理:
以及参与本网如下专文的相关内容:
医疗干预
一般治疗措施包括如下:
- 抗抑郁药物治疗:
- 可能对重度抑郁症患者是最有效的,这些药物可服用2-6周达到最大效果。
- 有许多不同类型的药物用来治疗抑郁症,医生将与患者一道筛选出副作用小、疗效最佳的药物和剂量。
- 其他治疗包括如:
- 电惊醒疗法(ECT)
- 迷走神经刺激疗法(VNS)
- 磁刺激疗法(TMS)
- 光疗法
心理疗法
对抑郁症的心理治疗由各种类型的咨询组成,如:
- 认知行为疗法(CBT)
- 人际心理治疗
- 心理动力学治疗
预防
预防患抑郁症的策略,包括如下:- 知晓到对本人的那些危险因素
- 如果需要,应做精神评估和心理治疗·
- 发展社会支持,如建立人际关系
- 学习压力管理技巧
- 定期锻炼
- 不滥用酒精或毒品
- 获得充足的睡眠、休息和娱乐
参考文献:
1. Zárate R et al. Significance of long chain polyunsaturated fatty acids in human health. Clin Transl Med. 2017;6(1):25.
2. Yang B et al. Clinical Efficacy and Biological Regulations of ω-3 PUFA-Derived Endocannabinoids in Major Depressive Disorder. Psychotherapy and psychosomatics. 2019;88(4):215-224.
3. Messamore E et al. Polyunsaturated fatty acids and recurrent mood disorders: Phenomenology, mechanisms, and clinical application. Prog Lipid Res. 2017;66:1-13.
4. Yu JZ et al. N-3 polyunsaturated fatty acids promote astrocyte differentiation and neurotrophin production independent of cAMP in patient-derived neural stem cells. Molecular psychiatry. 2021 Sep;26(9):4605-4615.
5. Grosso G et al. Dietary n-3 PUFA, fish consumption and depression: A systematic review and meta-analysis of observational studies. J Affect Disord. 2016;205:269-281.
6. Berger M et al. Relationship Between Polyunsaturated Fatty Acids and Psychopathology in the NEURAPRO Clinical Trial. Frontiers in psychiatry. 2019;10:393.
7. Scola G et al. Alterations in peripheral fatty acid composition in bipolar and unipolar depression. J Affect Disord. 2018;233:86-91.
8. Liao Y et al. Efficacy of omega-3 PUFAs in depression: A meta-analysis. Translational psychiatry. 2019;9(1):190.
9. Bozzatello P et al. Supplementation with Omega-3 Fatty Acids in Psychiatric Disorders: A Review of Literature Data. J Clin Med. 2016;5(8).
10. Keshavarz SA et al. Omega-3 supplementation effects on body weight and depression among dieter women with co-morbidity of depression and obesity compared with the placebo: A randomized clinical trial. Clin Nutr ESPEN. 2018;25:37-43.
11. Mazaherioun M et al. Long Chain n-3 Fatty Acids Improve Depression Syndrome in Type 2 Diabetes Mellitus. Iranian journal of public health. 2018;47(4):575-583.
12. Chang JP et al. Omega-3 polyunsaturated fatty acids in cardiovascular diseases comorbid major depressive disorder - Results from a randomized controlled trial. Brain Behav Immun. 2020;85:14-20.
13. Jiang W et al. Long-Chain Omega-3 Fatty Acid Supplements in Depressed Heart Failure Patients: Results of the OCEAN Trial. JACC Heart Fail. 2018;6(10):833-843.
14. Guu TW et al. International Society for Nutritional Psychiatry Research Practice Guidelines for Omega-3 Fatty Acids in the Treatment of Major Depressive Disorder. Psychotherapy and psychosomatics. 2019;88(5):263-273.
15. DiNicolantonio JJ et al. The Importance of Marine Omega-3s for Brain Development and the Prevention and Treatment of Behavior, Mood, and Other Brain Disorders. Nutrients. 2020;12(8).
16. Prasad AS et al. Molecular Mechanisms of Zinc as a Pro-Antioxidant Mediator: Clinical Therapeutic Implications. Antioxidants (Basel, Switzerland). 2019;8(6).
17. Doboszewska U et al. Zinc in the Monoaminergic Theory of Depression: Its Relationship to Neural Plasticity. Neural plasticity. 2017;2017:3682752.
18. Wang J et al. Magnesium, Selenium and Depression: A Review of the Evidence, Potential Mechanisms and Implications. Nutrients. 2018;10(5).
19. Petrilli MA et al. The Emerging Role for Zinc in Depression and Psychosis. Frontiers in pharmacology. 2017;8:414.
20. Li Z et al. Dietary zinc and iron intake and risk of depression: A meta-analysis. Psychiatry research. 2017;251:41-47.
21. Siwek M et al. Serum zinc level in depressed patients during zinc supplementation of imipramine treatment. J Affect Disord. 2010;126(3):447-452.
22. Szewczyk B et al. The role of magnesium and zinc in depression: similarities and differences. Magnesium research. 2018;31(3):78-89.
23. Solati Z et al. Zinc monotherapy increases serum brain-derived neurotrophic factor (BDNF) levels and decreases depressive symptoms in overweight or obese subjects: a double-blind, randomized, placebo-controlled trial. Nutritional neuroscience. 2015;18(4):162-168.
24. Sawada T et al. Effect of zinc supplementation on mood states in young women: a pilot study. European journal of clinical nutrition. 2010;64(3):331-333.
25. Ranjbar E et al. Effects of zinc supplementation on efficacy of antidepressant therapy, inflammatory cytokines, and brain-derived neurotrophic factor in patients with major depression. Nutritional neuroscience. 2014;17(2):65-71.
26. Nowak G et al. Effect of zinc supplementation on antidepressant therapy in unipolar depression: a preliminary placebo-controlled study. Pol J Pharmacol. 2003;55(6):1143-1147.
27. Siwek M, Dudek D, Paul IA, et al. Zinc supplementation augments efficacy of imipramine in treatment resistant patients: a double blind, placebo-controlled study. J Affect Disord. 2009;118(1-3):187-195.
28. Woodward G et al. Serum Vitamin D and Magnesium levels in a psychiatric cohort. Psychiatr Danub. 2019;31(Suppl 3):221-226.
29. Schaad KA et al. The relationship between vitamin D status and depression in a tactical athlete population. Journal of the International Society of Sports Nutrition. 2019;16(1):40.
30. Geng C et al. Vitamin D and depression: mechanisms, determination and application. Asia Pac J Clin Nutr. 2019;28(4):689-694.
31. Kim SY et al. Vitamin D deficiency and suicidal ideation: A cross-sectional study of 157,211 healthy adults. Journal of psychosomatic research. 2020;134:110125.
32. Gokalp G. The association between low vitamin D levels and suicide attempts in adolescents. Ann Clin Psychiatry. 2020;32(2):106-113.
33. Belzeaux R et al. Association between hypovitaminosis D and cognitive inhibition impairment during major depression episode. J Affect Disord. 2018;225:302-305.
34. Grudet C et al. Vitamin D and inflammation in major depressive disorder. J Affect Disord. 2020;267:33-41.
35. Okereke OI et al. Effect of Long-term Vitamin D3 Supplementation vs Placebo on Risk of Depression or Clinically Relevant Depressive Symptoms and on Change in Mood Scores: A Randomized Clinical Trial. JAMA. 2020;324(5):471-480.
36. Vellekkatt F et al. Efficacy of vitamin D supplementation in major depression: A meta-analysis of randomized controlled trials. J Postgrad Med. 2019;65(2):74-80.
37. Jamilian H et al. The effects of vitamin D supplementation on mental health, and biomarkers of inflammation and oxidative stress in patients with psychiatric disorders: A systematic review and meta-analysis of randomized controlled trials. Progress in neuro-psychopharmacology & biological psychiatry. 2019;94:109651.
38. Omidian M et al. Effects of vitamin D supplementation on depressive symptoms in type 2 diabetes mellitus patients: Randomized placebo-controlled double-blind clinical trial. Diabetes Metab Syndr. 2019;13(4):2375-2380.
39. Kotb MA et al. Effect of vitamin D replacement on depression in multiple sclerosis patients. Mult Scler Relat Disord. 2019;29:111-117.
40. Du Y et al. Crosstalk between the microbiota-gut-brain axis and depression. Heliyon. 2020;6(6):e04097.
41. Kim CS et al. Probiotic food consumption is associated with lower severity and prevalence of depression: A nationwide cross-sectional study. Nutrition (Burbank, Los Angeles County, Calif). 2019;63-64:169-174.
42. Sanada K et al. Gut microbiota and major depressive disorder: A systematic review and meta-analysis. J Affect Disord. 2020;266:1-13.
43. Chao L et al. Effects of Probiotics on Depressive or Anxiety Variables in Healthy Participants Under Stress Conditions or With a Depressive or Anxiety Diagnosis: A Meta-Analysis of Randomized Controlled Trials. Front Neurol. 2020;11:421.
44. Amirani E et al. The effects of probiotic supplementation on mental health, biomarkers of inflammation and oxidative stress in patients with psychiatric disorders: A systematic review and meta-analysis of randomized controlled trials. Complementary therapies in medicine. 2020;49:102361.
45. Yong SJ et al. Antidepressive Mechanisms of Probiotics and Their Therapeutic Potential. Front Neurosci. 2019;13:1361.
46. Goh KK et al. Effect of probiotics on depressive symptoms: A meta-analysis of human studies. Psychiatry research. 2019;282:112568.
47. Kazemi A et al. Effect of probiotic and prebiotic vs placebo on psychological outcomes in patients with major depressive disorder: A randomized clinical trial. Clin Nutr. 2019;38(2):522-528.
48. Messaoudi M et al. Assessment of psychotropic-like properties of a probiotic formulation (Lactobacillus helveticus R0052 and Bifidobacterium longum R0175) in rats and human subjects. The British journal of nutrition. 2011;105(5):755-764.
49. Saccarello A et al. Oral Administration of S-Adenosylmethionine (SAMe) and Lactobacillus Plantarum HEAL9 Improves the Mild-To-Moderate Symptoms of Depression: A Randomized, Double-Blind, Placebo-Controlled Study. Prim Care Companion CNS Disord. 2020;22(4).
50. Akkasheh G et al. Clinical and metabolic response to probiotic administration in patients with major depressive disorder: A randomized, double-blind, placebo-controlled trial. Nutrition. 2016;32(3):315-320.
51. Miyaoka T et al. Clostridium butyricum MIYAIRI 588 as Adjunctive Therapy for Treatment-Resistant Major Depressive Disorder: A Prospective Open-Label Trial. Clin Neuropharmacol. 2018;41(5):151-155.
52. Bambling M et al. A combination of probiotics and magnesium orotate attenuate depression in a small SSRI resistant cohort: an intestinal anti-inflammatory response is suggested. Inflammopharmacol. 2017;25(2):271-274.
53. Raygan F et al. The effects of probiotic and selenium co-supplementation on mental health parameters and metabolic profiles in type 2 diabetic patients with coronary heart disease: A randomized, double-blind, placebo-controlled trial. Clin Nutr. 2019;38(4):1594-1598.
54. Moludi J et al. The Effect of Probiotic Supplementation on Depressive Symptoms and Quality of Life in Patients After Myocardial Infarction: Results of a Preliminary Double-Blind Clinical Trial. Psychosomatic medicine. 2019;81(9):770-777.
55. Zhang L et al. Clinical characteristic and fecal microbiota responses to probiotic or antidepressant in patients with diarrhea-predominant irritable bowel syndrome with depression comorbidity: a pilot study. Chin Med J (Engl). 2019;132(3):346-351.
56. Pinto-Sanchez MI et al. Probiotic Bifidobacterium longum NCC3001 Reduces Depression Scores and Alters Brain Activity: A Pilot Study in Patients With Irritable Bowel Syndrome. Gastroenterology. 2017;153(2):448-459.e448.
57. Yang J et al. Landscapes of bacterial and metabolic signatures and their interaction in major depressive disorders. Science advances. 2020;6(49).
58. Mukherjee A et al. Gut microbes from the phylogenetically diverse genus Eubacterium and their various contributions to gut health. Gut Microbes. 2020;12(1):1802866.
59. Pennisi M et al. Acetyl-L-Carnitine in Dementia and Other Cognitive Disorders: A Critical Update. Nutrients. 2020;12(5).
60. Maldonado C et al. Potential Therapeutic Role of Carnitine and Acetylcarnitine in Neurological Disorders. Curr Pharm Des. 2020;26(12):1277-1285.
61. Wang SM et al. A review of current evidence for acetyl-l-carnitine in the treatment of depression. Journal of psychiatric research. 2014;53:30-37.
62. Nasca C et al. Acetyl-l-carnitine deficiency in patients with major depressive disorder. Proc Natl Acad Sci U S A. 2018;115(34):8627-8632.
63. Veronese N et al. Acetyl-L-Carnitine Supplementation and the Treatment of Depressive Symptoms: A Systematic Review and Meta-Analysis. Psychosomatic medicine. 2018;80(2):154-159.
64. Bersani G et al. L-Acetylcarnitine in dysthymic disorder in elderly patients: a double-blind, multicenter, controlled randomized study vs. fluoxetine. Eur Neuropsychopharmacol. 2013;23(10):1219-1225.
65. Leombruni P et al. A randomised controlled trial comparing duloxetine and acetyl L-carnitine in fibromyalgic patients: preliminary data. Clin Exp Rheumatol. 2015;33(1 Suppl 88):S82-85.
66. Rossini M et al. Double-blind, multicenter trial comparing acetyl l-carnitine with placebo in the treatment of fibromyalgia patients. Clin Exp Rheumatol. 2007;25(2):182-188.
67. Tardy AL et al. Vitamins and Minerals for Energy, Fatigue and Cognition: A Narrative Review of the Biochemical and Clinical Evidence. Nutrients. 2020;12(1).
68. Mikkelsen K et al. The Effects of Vitamin B in Depression. Curr Med Chem. 2016;23(38):4317-4337.
69. Zaric BL et al. Homocysteine and Hyperhomocysteinaemia. Curr Med Chem. 2019;26(16):2948-2961.
70. Young LM et al. A Systematic Review and Meta-Analysis of B Vitamin Supplementation on Depressive Symptoms, Anxiety, and Stress: Effects on Healthy and 'At-Risk' Individuals. Nutrients. 2019;11(9).
71. Almeida OP et al. Systematic review and meta-analysis of randomized placebo-controlled trials of folate and vitamin B12 for depression. International psychogeriatrics. 2015;27(5):727-737.
72. Mech AW et al. Correlation of clinical response with homocysteine reduction during therapy with reduced B vitamins in patients with MDD who are positive for MTHFR C677T or A1298C polymorphism: a randomized, double-blind, placebo-controlled study. The Journal of clinical psychiatry. 2016;77(5):668-671.
73. Tsujita N et al. Effect of Tryptophan, Vitamin B(6), and Nicotinamide-Containing Supplement Loading between Meals on Mood and Autonomic Nervous System Activity in Young Adults with Subclinical Depression: A Randomized, Double-Blind, and Placebo-Controlled Study. J Nutr Sci Vitaminol (Tokyo). 2019;65(6):507-514.
74. Roberts E et al. Caveat emptor: Folate in unipolar depressive illness, a systematic review and meta-analysis. Journal of psychopharmacology (Oxford, England). 2018;32(4):377-384.
75. Martone G. Enhancement of recovery from mental illness with l-methylfolate supplementation. Perspect Psychiatr Care. 2018;54(2):331-334.
76. Syed EU et al. Vitamin B12 supplementation in treating major depressive disorder: a randomized controlled trial. Open Neurol J. 2013;7:44-48.
77. Ghaleiha A et al. Adjuvant thiamine improved standard treatment in patients with major depressive disorder: results from a randomized, double-blind, and placebo-controlled clinical trial. European archives of psychiatry and clinical neuroscience. 2016;266(8):695-702.
78. Klinge CM et al. Dehydroepiandrosterone Research: Past, Current, and Future. Vitamins and hormones. 2018;108:1-28.
79. Stárka L et al. Dehydroepiandrosterone: a neuroactive steroid. J Steroid Biochem Mol Biol. 2015;145:254-260.
80. Hu Q et al. Clinical significance of decreased protein expression of dehydroepiandrosterone sulfate in the development of depression: a meta-analysis. J Affect Disord. 2015;174:416-423.
81. Mocking RJ et al. DHEAS and cortisol/DHEAS-ratio in recurrent depression: State, or trait predicting 10-year recurrence? Psychoneuroendocrinology. 2015;59:91-101.
82. Bloch M et al. Dehydroepiandrosterone treatment of midlife dysthymia. Biological psychiatry. 1999;45(12):1533-1541.
83. Schmidt PJ et al. Dehydroepiandrosterone monotherapy in midlife-onset major and minor depression. Archives of general psychiatry. 2005;62(2):154-162.
84. Wolkowitz OM et al. Double-blind treatment of major depression with dehydroepiandrosterone. The American journal of psychiatry. 1999;156(4):646-649.
85. Strous RD et al. Dehydroepiandrosterone augmentation in the management of negative, depressive, and anxiety symptoms in schizophrenia. Arch Gen Psychiatry. 2003;60(2):133-141.
86. Rabkin JG et al. Placebo-controlled trial of dehydroepiandrosterone (DHEA) for treatment of nonmajor depression in patients with HIV/AIDS. The American journal of psychiatry. 2006;163(1):59-66.
87. Jones LA et al. The ever-changing roles of serotonin. Int J Biochem Cell Biol. 2020:105776.
88. Kałużna-Czaplińska J et al. How important is tryptophan in human health? Crit Rev Food Sci Nutr. 2019;59(1):72-88.
89. Friedman M. Analysis, Nutrition, and Health Benefits of Tryptophan. Int J Tryptophan Res. 2018;11:1178646918802282.
90. Savitz J. Role of Kynurenine Metabolism Pathway Activation in Major Depressive Disorders. Curr Top Behav Neurosci. 2017;31:249-267.
91. Strasser B et al. Mood, food, and cognition: role of tryptophan and serotonin. Current opinion in clinical nutrition and metabolic care. 2016;19(1):55-61.
92. Ramírez LA et al. A new theory of depression based on the serotonin/kynurenine relationship and the hypothalamicpituitary- adrenal axis. Biomedica. 2018;38(3):437-450.
93. Dell'Osso L et al. Depression, Serotonin and Tryptophan. Curr Pharm Des. 2016;22(8):949-954.
94. Ramos-Chávez LA et al. Low Serum Tryptophan Levels as an Indicator of Global Cognitive Performance in Nondemented Women over 50 Years of Age. Oxid Med Cell Longev. 2018;2018:8604718.
95. Ogawa S et al. Plasma L-tryptophan concentration in major depressive disorder: new data and meta-analysis. The Journal of clinical psychiatry. 2014;75(9):e906-915.
96. Lieberman HR et al. Tryptophan Intake in the US Adult Population Is Not Related to Liver or Kidney Function but Is Associated with Depression and Sleep Outcomes. J Nutr. 2016;146(12):2609s-2615s.
97. Lindseth G et al. The effects of dietary tryptophan on affective disorders. Arch Psychiatr Nurs. 2015;29(2):102-107.
98. Kikuchi AM et al. A systematic review of the effect of L-tryptophan supplementation on mood and emotional functioning. J Diet Suppl. 2020:1-18.
99. Young SN. The effect of raising and lowering tryptophan levels on human mood and social behaviour. Philosophical transactions of the Royal Society of London Series B, Biological sciences. 2013;368(1615):20110375.
100. Gibson EL. Tryptophan supplementation and serotonin function: genetic variations in behavioural effects. The Proceedings of the Nutrition Society. 2018;77(2):174-188.
101. Jangid P et al. Comparative study of efficacy of l-5-hydroxytryptophan and fluoxetine in patients presenting with first depressive episode. Asian J Psychiatr. 2013;6(1):29-34.
102. Birdsall TC. 5-Hydroxytryptophan: a clinically-effective serotonin precursor. Altern Med Rev. 1998;3(4):271-280.
103. Jacobsen JPR et al. Adjunctive 5-Hydroxytryptophan Slow-Release for Treatment-Resistant Depression: Clinical and Preclinical Rationale. Trends Pharmacol Sci. 2016;37(11):933-944.
104. Das YT et al. Safety of 5-hydroxy-L-tryptophan. Toxicol Lett. 2004;150(1):111-122.
105. Deepmala et al. Clinical trials of N-acetylcysteine in psychiatry and neurology: A systematic review. Neuroscience and biobehavioral reviews. 2015;55:294-321.
106. Berk M et al. The efficacy of adjunctive N-acetylcysteine in major depressive disorder: a double-blind, randomized, placebo-controlled trial. The Journal of clinical psychiatry. 2014;75(6):628-636.
107. Porcu M et al. Effects of adjunctive N-acetylcysteine on depressive symptoms: Modulation by baseline high-sensitivity C-reactive protein. Psychiatry research. 2018;263:268-274.
108. Cullen KR et al. N-Acetylcysteine for Nonsuicidal Self-Injurious Behavior in Adolescents: An Open-Label Pilot Study. J Child Adolesc Psychopharmacol. 2018;28(2):136-144.
109. Fernandes BS et al. N-Acetylcysteine in depressive symptoms and functionality: a systematic review and meta-analysis. J Clin Psychiatry. 2016;77(4):e457-466.
110. Bottiglieri T. Folate, vitamin B₁₂, and S-adenosylmethionine. Psychiatr Clin North Am. 2013 Mar;36(1):1-13.
111. Sharma A et al. S-Adenosylmethionine (SAMe) for Neuropsychiatric Disorders: A Clinician-Oriented Review of Research. J Clin Psychiatry. 2017;78(6):e656-e667.
112. Williams AL et al. S-adenosylmethionine (SAMe) as treatment for depression: a systematic review. Clinical and investigative medicine Medecine clinique et experimentale. 2005;28(3):132-139.
113. Bressa GM. S-adenosyl-l-methionine (SAMe) as antidepressant: meta-analysis of clinical studies. Acta Neurol Scand Suppl. 1994;154:7-14.
114. Hardy ML et al. S-adenosyl-L-methionine for treatment of depression, osteoarthritis, and liver disease. Evid Rep Technol Assess (Summ). 2003(64):1-3.
115. Sarris J et al. S-Adenosylmethionine (SAMe) monotherapy for depression: an 8-week double-blind, randomised, controlled trial. Psychopharmacology. 2020;237(1):209-218.
116. Sarris J et al. Adjunctive S-adenosylmethionine (SAMe) in treating non-remittent major depressive disorder: An 8-week double-blind, randomized, controlled trial<sup/>. Eur Neuropsychopharmacol. 2018;28(10):1126-1136.
117. Galizia I et al. S-adenosyl methionine (SAMe) for depression in adults. The Cochrane database of systematic reviews. 2016;10(10):Cd011286.
118. Mischoulon D et al. A double-blind, randomized, placebo-controlled clinical trial of S-adenosyl-L-methionine (SAMe) versus escitalopram in major depressive disorder. The Journal of clinical psychiatry. 2014;75(4):370-376.
119. Sarris J et al. Is S-Adenosyl Methionine (SAMe) for Depression Only Effective in Males? A Re-Analysis of Data from a Randomized Clinical Trial. Pharmacopsychiatry. 2015;48(4-5):141-144.
120. Karas Kuželički N. S-Adenosyl Methionine in the Therapy of Depression and Other Psychiatric Disorders. Drug Dev Res. 2016;77(7):346-356.
121. Kious BM et al. Creatine for the Treatment of Depression. Biomolecules. 2019;9(9).
122. Bakian AV et al. Dietary creatine intake and depression risk among U.S. adults. Translational psychiatry. 2020;10(1):52.
123. Lyoo IK et al. A randomized, double-blind placebo-controlled trial of oral creatine monohydrate augmentation for enhanced response to a selective serotonin reuptake inhibitor in women with major depressive disorder. The American journal of psychiatry. 2012;169(9):937-945.
124. Yoon S et al. Effects of Creatine Monohydrate Augmentation on Brain Metabolic and Network Outcome Measures in Women With Major Depressive Disorder. Biological psychiatry. 2016;80(6):439-447.
125. Kious BM et al. An Open-Label Pilot Study of Combined Augmentation With Creatine Monohydrate and 5-Hydroxytryptophan for Selective Serotonin Reuptake Inhibitor- or Serotonin-Norepinephrine Reuptake Inhibitor-Resistant Depression in Adult Women. Journal of clinical psychopharmacology. 2017;37(5):578-583.
126. Kondo DG et al. Open-label adjunctive creatine for female adolescents with SSRI-resistant major depressive disorder: a 31-phosphorus magnetic resonance spectroscopy study. J Affect Disord. 2011;135(1-3):354-361.
127. Nemets B et al. A pilot dose-finding clinical trial of creatine monohydrate augmentation to SSRIs/SNRIs/NASA antidepressant treatment in major depression. International clinical psychopharmacology. 2013;28(3):127-133.
128. Colodny L et al. Inositol--clinical applications for exogenous use. Alternative medicine review : a journal of clinical therapeutic. 1998;3(6):432-447.
129. Zheng H et al. High-frequency rTMS treatment increases left prefrontal myo-inositol in young patients with treatment-resistant depression. Progress in neuro-psychopharmacology & biological psychiatry. 2010;34(7):1189-1195.
130. Moore CM et al. Effects of myo-inositol ingestion on human brain myo-inositol levels: a proton magnetic resonance spectroscopic imaging study. Biological psychiatry. 1999;45(9):1197-1202.
131. Levine J et al. Double-blind, controlled trial of inositol treatment of depression. The American journal of psychiatry. 1995;152(5):792-794.
132. Mukai T, Kishi T, Matsuda Y, Iwata N. A meta-analysis of inositol for depression and anxiety disorders. Human psychopharmacology. 2014;29(1):55-63.
133. Levine J et al. Combination of inositol and serotonin reuptake inhibitors in the treatment of depression. Biological psychiatry. 1999;45(3):270-273.
134. Nemets B et al. Inositol addition does not improve depression in SSRI treatment failures. Journal of neural transmission (Vienna, Austria : 1996). 1999;106(7-8):795-798.
135. Botturi A et al. The Role and the Effect of Magnesium in Mental Disorders: A Systematic Review. Nutrients. 2020;12(6).
136. Tarleton EK et al. The Association between Serum Magnesium Levels and Depression in an Adult Primary Care Population. Nutrients. 2019;11(7).
137. Sun C et al. Dietary magnesium intake and risk of depression. J Affect Disord. 2019;246:627-632.
138. Phelan D et al. Magnesium and mood disorders: systematic review and meta-analysis. BJPsych Open. 2018;4(4):167-179.
139. Tarleton EK et al. Role of magnesium supplementation in the treatment of depression: A randomized clinical trial. PLoS One. 2017;12(6):e0180067.
140. Rajizadeh A et al. Effect of magnesium supplementation on depression status in depressed patients with magnesium deficiency: A randomized, double-blind, placebo-controlled trial. Nutrition (Burbank, Los Angeles County, Calif). 2017;35:56-60.
141. Vink R. Magnesium in the CNS: recent advances and developments. Magnesium research. 2016;29(3):95-101.
142. Sun Q et al. Regulation of structural and functional synapse density by L-threonate through modulation of intraneuronal magnesium concentration. Neuropharmacology. 2016;108:426-439.
143. Kim YS et al. Neuroprotective effects of magnesium L-threonate in a hypoxic zebrafish model. BMC neuroscience. 2020;21(1):29.
144. Shen Y et al. Treatment Of Magnesium-L-Threonate Elevates The Magnesium Level In The Cerebrospinal Fluid And Attenuates Motor Deficits And Dopamine Neuron Loss In A Mouse Model Of Parkinson's disease. Neuropsychiatr Dis Treat. 2019;15:3143-3153.
145. Jia S et al. Elevation of Brain Magnesium Potentiates Neural Stem Cell Proliferation in the Hippocampus of Young and Aged Mice. J Cell Physiol. 2016;231(9):1903-1912.
146. Sadir S et al. Neurobehavioral and biochemical effects of magnesium chloride (MgCl2), magnesium sulphate (MgSO4) and magnesium-L-threonate (MgT) supplementation in rats: A dose dependent comparative study. Pakistan journal of pharmaceutical sciences. 2019;32(1(Supplementary)):277-283.
147. Ochoa-de la Paz L et al. Taurine and GABA neurotransmitter receptors, a relationship with therapeutic potential? Expert Rev Neurother. 2019;19(4):289-291.
148. Cui T et al. Abnormal levels of seven amino neurotransmitters in depressed rat brain and determination by HPLC-FLD. Biomed Chromatogr. 2017;31(8).
149. Caletti G et al. Antidepressant effect of taurine in diabetic rats. Amino Acids. 2012;43(4):1525-1533.
150. Caletti G et al. Antidepressant dose of taurine increases mRNA expression of GABAA receptor α2 subunit and BDNF in the hippocampus of diabetic rats. Behavioural brain research. 2015;283:11-15.
151. Wu GF et al. Antidepressant effect of taurine in chronic unpredictable mild stress-induced depressive rats. Sci Rep. 2017;7(1):4989.
152. O'Donnell CP et al. Adjunctive Taurine in First-Episode Psychosis: A Phase 2, Double-Blind, Randomized, Placebo-Controlled Study. The Journal of clinical psychiatry. 2016;77(12):e1610-e1617.
153. Asher GN et al. Comparative Benefits and Harms of Complementary and Alternative Medicine Therapies for Initial Treatment of Major Depressive Disorder: Systematic Review and Meta-Analysis. Journal of alternative and complementary medicine (New York, NY). 2017;23(12):907-919.
154. Kasper S et al. Better tolerability of St. John's wort extract WS 5570 compared to treatment with SSRIs: a reanalysis of data from controlled clinical trials in acute major depression. Int Clin Psychopharmacol. 2010;25(4):204-213.
155. Freeman MP et al. Complementary and alternative medicine for major depressive disorder: a meta-analysis of patient characteristics, placebo-response rates, and treatment outcomes relative to standard antidepressants. The Journal of clinical psychiatry. 2010;71(6):682-688.
156. Apaydin EA et al. A systematic review of St. John's wort for major depressive disorder. Syst Rev. 2016;5(1):148.
157. Herraiz T et al. Monoamine Oxidase-A Inhibition and Associated Antioxidant Activity in Plant Extracts with Potential Antidepressant Actions. Biomed Res Int. 2018;2018:4810394.
158. Sevastre-Berghian AC et al. Characterization and biological effects of Hypericum extracts on experimentally-induced - anxiety, oxidative stress and inflammation in rats. Journal of physiology and pharmacology : an official journal of the Polish Physiological Society. 2018;69(5).
159. Schmidt M et al. The mechanisms of action of St. John's wort: an update. Wien Med Wochenschr. 2015;165(11-12):229-235.
160. Nicolussi S et al. Clinical relevance of St. John's wort drug interactions revisited. Br J Pharmacol. 2020;177(6):1212-1226.
161. Blumenthal M et al. Herbal Medicine Expanded Commission E Monographs. Dallas, TX: American Botanical Council; 2000.
162. Khazdair MR at al. The effects of Crocus sativus (saffron) and its constituents on nervous system: A review. Avicenna J Phytomed. 2015;5(5):376-391.
163. Dai L et al. Safety and Efficacy of Saffron (Crocus sativus L.) for Treating Mild to Moderate Depression: A Systematic Review and Meta-analysis. The Journal of nervous and mental disease. 2020.
164. Khaksarian M et al. The efficacy of Crocus sativus (Saffron) versus placebo and Fluoxetine in treating depression: a systematic review and meta-analysis. Psychology research and behavior management. 2019;12:297-305.
165. Tóth B et al. The Efficacy of Saffron in the Treatment of Mild to Moderate Depression: A Meta-analysis. Planta Med. 2019;85(1):24-31.
166. Yang X et al. Comparative efficacy and safety of Crocus sativus L. for treating mild to moderate major depressive disorder in adults: a meta-analysis of randomized controlled trials. Neuropsychiatr Dis Treat. 2018;14:1297-1305.
167. Ghaderi A et al. The effects of saffron (Crocus sativus L.) on mental health parameters and C-reactive protein: A meta-analysis of randomized clinical trials. Complement Ther Med. 2020;48:102250.
168. Talaei A et al. Crocin, the main active saffron constituent, as an adjunctive treatment in major depressive disorder: a randomized, double-blind, placebo-controlled, pilot clinical trial. J Affect Disord. 2015;174:51-56.
169. Milajerdi A et al. The effects of alcoholic extract of saffron (Crocus satious L.) on mild to moderate comorbid depression-anxiety, sleep quality, and life satisfaction in type 2 diabetes mellitus: A double-blind, randomized and placebo-controlled clinical trial. Complement Ther Med. 2018;41:196-202.
170. Lopresti AL et al. Efficacy of a standardised saffron extract (affron®) as an add-on to antidepressant medication for the treatment of persistent depressive symptoms in adults: A randomised, double-blind, placebo-controlled study. Journal of psychopharmacology (Oxford, England). 2019;33(11):1415-1427.
171. Akhondzadeh S et al. A placebo controlled randomized clinical trial of Crocus sativus L. (saffron) on depression and food craving among overweight women with mild to moderate depression. J Clin Pharm Ther. 2020;45(1):134-143.
172. Kashani L et al. Efficacy of Crocus sativus (saffron) in treatment of major depressive disorder associated with post-menopausal hot flashes: a double-blind, randomized, placebo-controlled trial. Arch Gynecol Obstet. 2018;297(3):717-724.
173. Jalali F et al. The Effect of Saffron on Depression among Recovered Consumers of Methamphetamine Living with HIV/AIDS. Subst Use Misuse. 2018;53(12):1951-1957.
174. Shahmansouri N et al. A randomized, double-blind, clinical trial comparing the efficacy and safety of Crocus sativus L. with fluoxetine for improving mild to moderate depression in post percutaneous coronary intervention patients. J Affect Disord. 2014;155:216-222.
175. Shakiba M et al. Saffron (Crocus sativus) versus duloxetine for treatment of patients with fibromyalgia: A randomized double-blind clinical trial. Avicenna journal of phytomedicine. 2018;8(6):513-523.
176. Kashani L et al. Saffron for treatment of fluoxetine-induced sexual dysfunction in women: randomized double-blind placebo-controlled study. Human psychopharmacology. 2013;28(1):54-60.
177. Modabbernia A et al. Effect of saffron on fluoxetine-induced sexual impairment in men: randomized double-blind placebo-controlled trial. Psychopharmacology (Berl). 2012;223(4):381-388.
178. Kim M et al. Effects of Lavender on Anxiety, Depression, and Physiological Parameters: Systematic Review and Meta-Analysis. Asian Nurs Res (Korean Soc Nurs Sci). 2021 Dec;15(5):279-290.
179. Bazrafshan MR et al. The effect of lavender herbal tea on the anxiety and depression of the elderly: A randomized clinical trial. Complementary therapies in medicine. 2020;50:102393.
180. Kasper S et al. Efficacy of Silexan in mixed anxiety-depression--A randomized, placebo-controlled trial. Eur Neuropsychopharmacol. 2016;26(2):331-340.
181. Uehleke B et al. Phase II trial on the effects of Silexan in patients with neurasthenia, post-traumatic stress disorder or somatization disorder. Phytomedicine. 2012;19(8-9):665-671.
182. Akhondzadeh S et al. Comparison of Lavandula angustifolia Mill. tincture and imipramine in the treatment of mild to moderate depression: a double-blind, randomized trial. Progress in neuro-psychopharmacology & biological psychiatry. 2003;27(1):123-127.
183. Ross SM. Rhodiola rosea (SHR-5), Part 2: A standardized extract of Rhodiola rosea is shown to be effective in the treatment of mild to moderate depression. Holistic nursing practice. 2014;28(3):217-221.
184. Limanaqi F et al. Potential Antidepressant Effects of Scutellaria baicalensis, Hericium erinaceus and Rhodiola rosea. Antioxidants (Basel, Switzerland). 2020;9(3).
185. Concerto C et al. Exploring the effect of adaptogenic Rhodiola Rosea extract on neuroplasticity in humans. Complement Ther Med. 2018;41:141-146.
186. Mao JJ et al. Rhodiola rosea versus sertraline for major depressive disorder: A randomized placebo-controlled trial. Phytomedicine. 2015;22(3):394-399.
187. Darbinyan V et al. Clinical trial of Rhodiola rosea L. extract SHR-5 in the treatment of mild to moderate depression. Nord J Psychiatry. 2007;61(5):343-348.
188. Gao L et al. Antidepressants effects of Rhodiola capsule combined with sertraline for major depressive disorder: A randomized double-blind placebo-controlled clinical trial. J Affect Disord. 2020;265:99-103.
189. Amsterdam JD et al. Rhodiola rosea L. as a putative botanical antidepressant. Phytomedicine. 2016;23(7):770-783.
190. Araj-Khodaei M et al. A double-blind, randomized pilot study for comparison of Melissa officinalis L. and Lavandula angustifolia Mill. with Fluoxetine for the treatment of depression. BMC Complement Med Ther. 2020;20(1):207-207.
191. López V et al. Neuroprotective and neurological properties of Melissa officinalis. Neurochem Res. 2009;34(11):1955-1961.
192. Kirsch I. Placebo Effect in the Treatment of Depression and Anxiety. Frontiers in psychiatry. 2019;10:407.
193. Haybar H et al. The effects of Melissa officinalis supplementation on depression, anxiety, stress, and sleep disorder in patients with chronic stable angina. Clinical nutrition ESPEN. 2018;26:47-52.
194. Ranjbar M et al. Effects of Herbal combination (Melissa officinalis L. and Nepeta menthoides Boiss. & Buhse) on insomnia severity, anxiety and depression in insomniacs: Randomized placebo controlled trial. Integr Med Res. 2018;7(4):328-332.
参考来源:
www.mayoclinic.org
美国抑郁和双相障碍支持联盟
http://www.dbsalliance.org
美国国家公众健康网
www.medlienplus.gov
美国心理健康网
http://www.mentalhealthamerica.net
加拿大心理健康协会
http://www.ontario.cmha.ca
加拿大精神病协会
http://www.cpa-apc.org
免责声明和安全信息
- 本信息(包括任何附带资料)不是为了取代医生或有关合格从业人士的建议或忠告。
- 任何人如果想要对本文涉及的药物、饮食、运动或其他生活方式的使用、或改变调整,以预防或治疗某一特定健康状况或疾病,应首先咨询医生或有关合格从业人士,并获得他/她们的许可。妊娠和哺乳妇女在使用本网站任何内容前,尤其应征求医生的意见。
- 除非另有说明,本网站所述内容仅适用于成人。
- 本网站所推荐的任何产品,消费者应该以实际的产品标签内容为准,尤其应关注重要的安全信息以及产品最新信息,包括剂量、使用方法和禁忌症等。
- 由于循证医学研究、文献及有关产品处于不断的变化中,本网站工作人员将尽力更新。
- 本网站不能保证所载文章内容、综合干预方案以及相关成分或产品述及的健康益处,也不承担任何责任。

